Weekly Tech+Bio #80: Organoids on Artemis II
Anthropic's $400M AI biotech acqui-hire, CRO stocks reprice, new EMA draft, and pharma's spring acquisition spree
Anthropic pays $400M for a 10-person biotech AI startup, pharma signs $25.5B in acquisitions over eight days, CRO stocks drop on the possibility that AI unbundles the intelligence layer from the execution layer, and bone marrow organoids are heading to the Moon.
Hi! This is BiopharmaTrend’s weekly newsletter, Where Tech Meets Bio, where we explore technologies, breakthroughs, and cutting-edge companies.
If this newsletter is in your inbox, it’s because you subscribed, or someone thought you might enjoy it. In either case, you can subscribe directly by clicking this button:
🤖 AI x Bio
(AI applications in drug discovery, biotech, and healthcare)
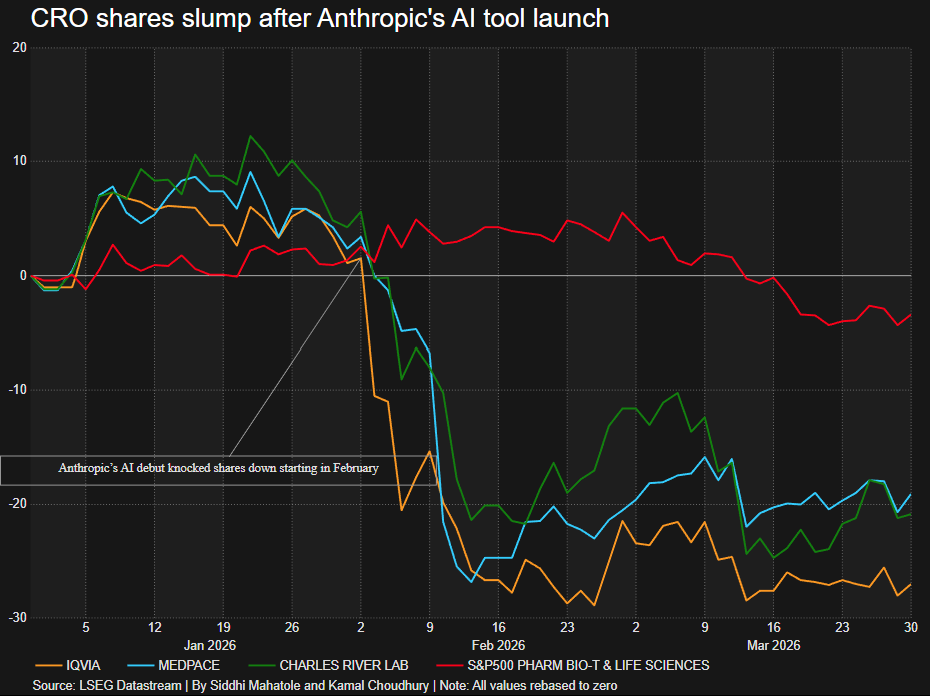
The CRO intelligence layer remains unclaimed
Reuters reported that CRO stocks (IQVIA, Medpace, Charles River) have dropped sharply since Anthropic launched AI agents in February. The authors’ conclusion is that the market is overreacting, AI can’t replace patient recruitment and site execution, CROs will be broadly fine.
The near-term read is probably right, but the longer-term question is more interesting: what happens when the design layer of trials (protocol optimization, site selection, patient matching) gets commoditized by AI tools that sponsors can run themselves?
As noted recently, strategic CROs sell execution and intelligence bundled together. Unbundle that, and they’re competing on operations and logistics alone, which is a different kind of business that’s priced differently.
There’s also a second angle: if AI lowers the intelligence barrier, a new class of leaner, tech-native CROs could assemble trial infrastructure on demand, more platform than services firm. This structural shift is explored in our 2023 BiopharmaTrend analysis of the CRO industry, though agentic capabilities at the time were still early. Nobody is cutting CRO spend yet. But “no evidence of cuts” is not the same as “no shift in leverage.”
Agentic AI in drug discovery is already delivering value, but not where the hype points
📝 A new review in Drug Discovery Today covers eight case studies from companies including Coincidence Labs, Human Chemical, Potato, Kiin Bio, Augmented Nature, onepot, Plex Research, and Convexia. Some reported specific quantitative benchmarks, not just “we used AI and it was faster.”
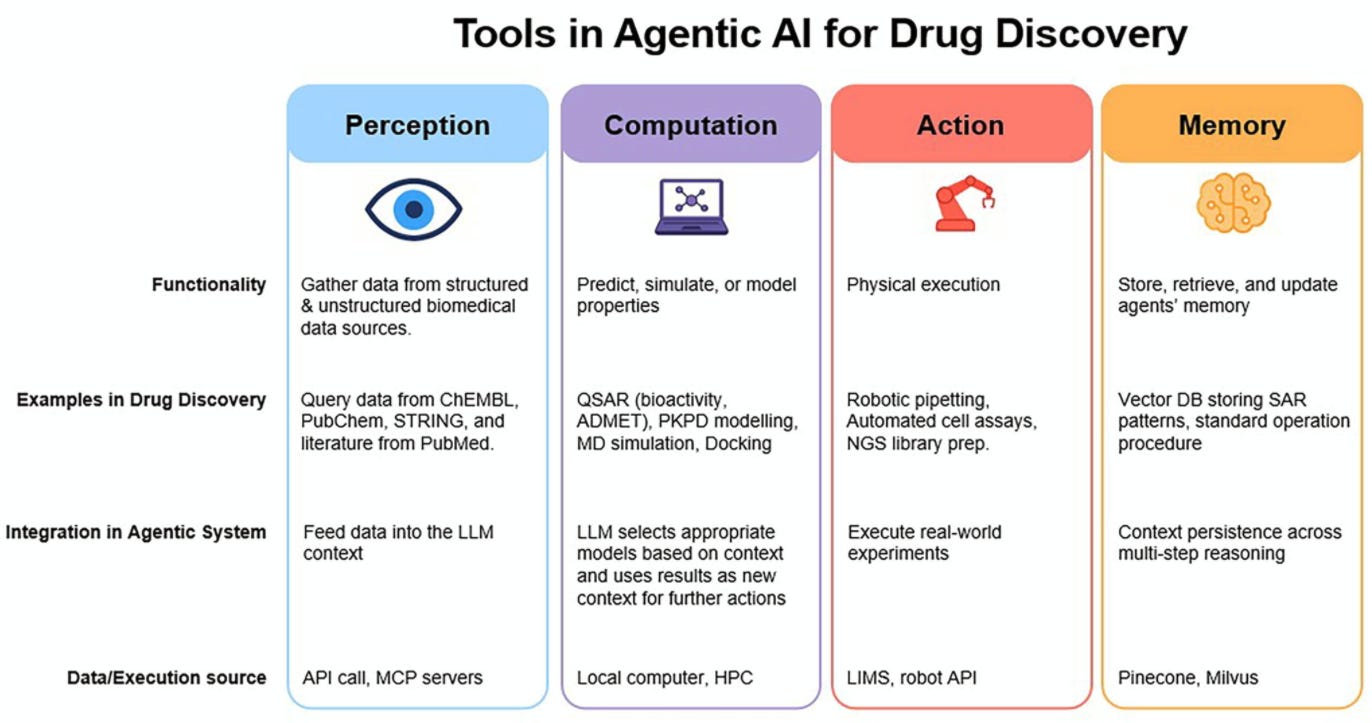
The most convincing gains are in the tedious middle: literature synthesis, protocol generation, assay design. Potato’s Tater agent took the design cycle for a qPCR assay from literature review through to executable lab automation code from 1-4 months to under two hours. Empirical validation was still needed, but the bottleneck it removes is the coordination tax between “we know what assay we need” and “here’s a draft protocol ready for the bench.”
Note: there’s missing cost data: every case study reports time savings and none report what the agentic infrastructure costs to build and maintain.
The authors flag that current benchmarks capture whether an agent got the right answer but not whether the reasoning behind it was sound. And a separate evaluation from Princeton and Cornell suggests the problem runs deeper: across 14 AI agent systems, agents that could solve a task often failed on repeated attempts under identical conditions. Bigger models didn’t reliably fix this.
Our read: the real value right now is compressing the overhead between steps that already work on their own, not autonomous science (yet). We went deeper on where agents are actually being deployed, what’s breaking, and what alternatives look like in our recent deep dive:
➕ Fay Lin at GEN also surveyed agentic platforms now on the life sciences market after NVIDIA GTC: Kosmos, LabOS, Latent-Y, Dyno Psi-Phi, and others.
Is AI replacing radiologists?
The “AI replacing radiologists” discourse resurfaced this week after NYC Health + Hospitals CEO Mitchell Katz said AI could replace a significant portion of radiology work—initial AI reads, radiologist review of flagged abnormals. The Stanford-Harvard NOHARM study showed top LLMs outperforming generalist physicians on clinical safety, and the numbers look good in controlled settings.
Bo Wang, Chief AI Scientist at University Health Network (Canada’s largest research hospital network), offered a corrective from deployment experience:
The accuracy is real in narrow tasks. But NOHARM found 76.6% of AI errors were omissions = things the model never flagged. In a hospital, a missed finding propagates: downstream physicians trust the AI read, the patient waits, the window closes. (One caveat: NOHARM evaluated LLMs on clinical recommendations across ten specialties, not imaging-specific AI, though the omission pattern maps to radiology concerns.)
No hospital system has solved the accountability question either. When an AI-assisted diagnosis causes harm, the liability chain between physician, hospital, and vendor has no established framework. Which is not regulatory delay but “...a fundamental gap in the infrastructure for AI-in-medicine.”
In Wang’s view, here’s what’s actually happening: AI is reshaping the radiology job, not eliminating it. Routine reads get faster, time shifts to complex cases and clinical correlation. The capability question is nearly answered, while the deployment question has barely been asked.
AI clinical operations partnerships keep stacking
🔹 Bristol Myers Squibb and Faro Health announced a multi-year deal to apply AI across clinical trial design and protocol workflows. Faro structures protocols into standardized, machine-readable formats and benchmarks them against datasets like Tufts CSDD, with work spanning both early- and late-phase programs. Faro is not an isolated instance—this year BMS also partnered with Evinova on trial design and with Microsoft on AI-based patient identification.
🔹Tempus reports that its ALERT trial, conducted with Medtronic across 35 U.S. hospitals, showed that AI-driven alerts embedded in EHRs improved detection and treatment of severe heart valve diseases. The study had 765 clinicians, over 2,000 echocardiograms, and used Tempus’s “Next” to analyze echo reports in real time with NLP and notify clinicians about high-risk findings.
🔹 OpenEvidence partnered with Tandem to connect real-time, AI-driven clinical decision support with automated prescription and insurance workflows. OpenEvidence functions as a clinical search layer, synthesizing evidence from sources like NEJM, JAMA, and Cochrane to support treatment decisions at the point of care, with over 1 million daily consultations in the U.S. Described as “ChatGPT for doctors,” OE raised $250M at a $12B valuation this year and generates over $100M in annual revenue, in part through ads embedded in the platform.
AI in the pipeline layer
🔹 Boehringer Ingelheim is expanding its collaboration with OpenProtein.AI to push AI-driven antibody discovery further into its development workflows. The pairing of a large pharma with deep therapeutic pipelines and a startup built around protein foundation models is becoming a common pattern.
🔹 Infinimmune and Merck entered a discovery and development partnership—Infinimmune will use its platform to screen human immune cells and identify naturally occurring antibodies, then refine them with AI tools. Merck gains exclusive commercialization rights, with potential milestone payments up to $838M across multiple undisclosed disease targets.
🔹 Evotec, Germany-based drug R&D company operating a large network of partnered R&D platforms, appointed Ashiq Khan as chief commercial officer, bringing AI and robotics experience from previous roles at Iktos and Schrödinger. The hire is part of Evotec’s broader transformation plan announced last month, which named commercial execution as a core focus.
🔹 Insilico Medicine partner TaiGen Biotechnology achieved first-subject enrollment and dosing in the Phase 1 trial of ISM4808—an oral HIF-PHD inhibitor for CKD-related anemia discovered with Insilico’s generative AI platform for small-molecule design in under three months (from the December 2025 in-licensing). Another data point for Insilico’s growing list of partnered programs moving into clinic.
🔹 Enveda Biosciences reported early clinical data from its Phase 1b trial of an oral candidate discovered via the company’s AI platform, in moderate-to-severe atopic dermatitis. Nine patients treated at 800 mg daily for 28 days showed rapid reductions in disease severity and itch within the first week, with improvements continuing after treatment cessation. No serious adverse events were reported. ENV-294 is a non-degrading stabilizer (LOCKTAC) showing biomarker activity across Th1, Th2, and Th17 pathways—broader modulation than single-cytokine-axis therapies. Enveda plans Phase 2a trials in atopic dermatitis and asthma, with Phase 2b targeted for mid-2026.
🔹 Lantern Pharma, the AI-driven oncology company known for its RADR AI platform, is joining the agentic wave with withZeta.ai—a multi-agentic co-scientist platform for rare cancer drug discovery that autonomously queries proprietary knowledge bases, clinical trial databases, and molecular libraries. The company will demo the platform at an investor briefing on April 9.
💰 Money Flows
(Funding rounds, IPOs, and M&A for startups and smaller companies)
Pharma went on a $25.5B, eight-day acquisition spree. Six pharmaceutical companies signed acquisitions worth up to $25.5B by the start of this month, including two $5B+ upfront deals disclosed on the same Tuesday morning: Eli Lilly’s $6.3B bet on Centessa Pharmaceuticals’ orexin drug candidates and Biogen’s acquisition of Apellis Pharmaceuticals. For context, there were a total of seven deals of that magnitude in all of 2025.
Across March, biopharmas lined up 10 acquisitions worth up to $31.5B, per Endpoints News. Eight included contingent value rights or milestone payments—a dealmaking structure that has become more common in recent years. Year-to-date, Endpoints has tracked up to $48B in total potential deal value across 19 biopharma acquisitions. Société Générale tallied $126B in total deal value for all of 2025, well above the 10-year average of $85B. If the current pace holds, 2026 could surpass it.
🔹 Blackstone has collected $6.3B for its latest life sciences fund, which has already committed roughly $2B over the past year for an ADC at Merck, an anti-TL1A at Teva-partnered Sanofi, and other deals. The fund invests in approximately 3% of deals it evaluates, targeting therapies with $1B+ peak sales potential.
🔹 Anthropic acquired Coefficient Bio, a stealth biotech AI startup, for $400M in stock. Coefficient Bio was founded eight months ago by Samuel Stanton and Nathan C. Frey, both from Genentech’s Prescient Design group. The team of roughly 10 people was using AI for drug discovery planning, clinical regulatory strategy, candidate identification, and biomolecule modeling. As Jonah Probell of Lexi Ventures noted, the deal generated a reported 38,513% paper IRR for Dimension, the VC firm that backed them. Anthropic’s head of Healthcare & Life Sciences Eric Kauderer-Abrams stated the goal rather plainly: Anthropic wants a meaningful share of all life science work globally to run on Claude. The team is expected to join Anthropic’s health and life sciences unit.
🔹 Generare, a Paris-based techbio building a discovery platform around microbial DNA, raised €20M in a Series A co-led by Alven and daphni. The company identifies biosynthetic gene clusters in microbial genomes, expresses them in lab systems, and characterizes the resulting molecules, reporting over 200 previously uncharacterized small molecules per cycle versus an industry baseline of ~45. Most screening libraries draw from an estimated 3% of accessible chemical space, and Generare is betting that new chemistry from unexplored microbial genomes, not just more data from known chemistry, is what AI-driven drug discovery needs. The team of ~25 aims to scale output to 2,000+ molecules by 2027. Founded in 2023 by Guillaume Vandenesch and Vincent Libis.
🔹 Scala Biodesign, an Israeli computational protein design platform, raised $16M in a Series A led by Grove Ventures, joined by TLV Partners, Deep Insight, and the Israel Innovation Authority. The platform combines LLMs, physics-based modeling, and evolutionary data to help clients design and optimize proteins for biologic, enzyme, antibody, and vaccine therapies. It aims to become routine infrastructure across the full development pipeline rather than a one-off discovery tool.
🏛️ Bioeconomy & Society
(News on centers, regulatory updates, and broader biotech ecosystem developments)
The EMA just issued its first draft qualification opinion for virtual control groups as a New Approach Methodology.
Context is rather narrow: rat dose-range finding studies, non-GLP, replacing concurrent control groups with virtual comparators built from historical data. It’s a narrow starting point, but it sets a regulatory blueprint for every study design where live controls exist by convention rather than scientific necessity.
Stefano Gaburro, who leads the Pistoia Alliance Minimal Metadata Set Working Group, noted that the real bottleneck was never the algorithm, but the metadata. Inconsistent annotation across institutions prevented historical control data from being pooled and reused. Pistoia’s MNMS framework, notes Stefano, was built specifically to create the data infrastructure that virtual control groups require. A cross-pharma proof of concept with five companies, using 24/7 home cage monitoring data, is ongoing. The EMA consultation runs until May 12, 2026.
Around the same time, Joseph Wu and colleagues published a review in Cell on NAMs for drug discovery, framing the evolution from 2D stem cell systems through 3D organoid models to AI-driven in silico platforms. The review argues that the next 30 years of drug development will see animal models transition from a central role to a supporting one, following the 3Rs principle, replaced increasingly by human-centric NAMs powered by multi-omics data and AI pipelines. Between the EMA opinion and this review, the regulatory and scientific architecture for reducing animal use in drug development is being built in parallel.
⚙️ Other Tech
(Innovations across quantum computing, BCIs, gene editing, and more)
🔹 Bone marrow by the moon—NASA’s Artemis II mission is carrying AVATAR (A Virtual Astronaut Tissue Analog Response)—organ-on-chip devices containing bone marrow cells from crew members to study how deep space radiation and microgravity affect human health during the approximately 10-day lunar journey.
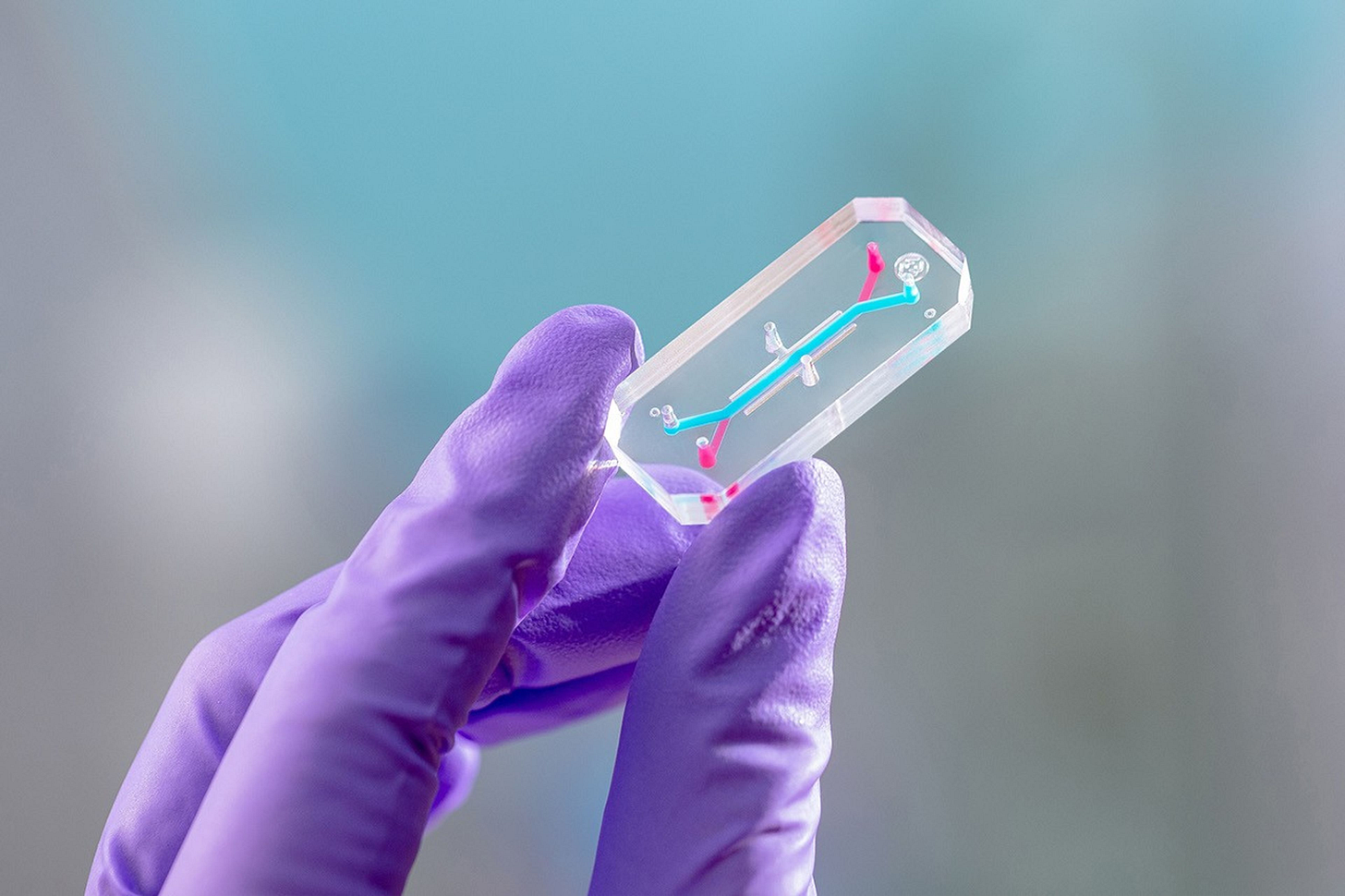
Bone marrow was selected for its radiosensitivity and centrality to immune function. The battery-powered payload, developed by Space Tango, will maintain automated environmental controls during flight. Upon return, Emulate researchers will use single-cell RNA sequencing to compare flight samples against ground-based controls. NASA has extended organ-on-chip viability to six months minimum to enable observation of long-term disease and treatment responses.
🚀 A New Kid on the Dura
Epia Neuro launched from San Francisco this week with a minimally invasive brain-computer interface designed to help stroke survivors regain motor function by reading what the brain intends to do and translating that into movement.
The founder is Michel Maharbiz, who previously built iota Biosciences, a bioelectronics startup later acquired by Astellas. The team spans engineering and neuroscience, with backgrounds in both research and clinical system development.
Stroke is the entry point: roughly 690,000 cases annually in the U.S., with around 60,000 patients potentially eligible for device-based intervention. Epia plans first-in-human demonstrations with the Department of Neurosurgery at Lenox Hill Hospital.
The technical differentiator is a read/write architecture where the device can both record brain signals and deliver stimulation, spanning cortical and deeper brain regions. This contrasts with earlier generations of primarily read-only BCIs. The implant is placed within the skull without penetrating brain tissue via a roughly one-hour surgery. Onboard algorithms adapt to each patient’s activity patterns over time, with wireless recharging and upgradeable architecture built in for long-term use.
Early on, the device supports rehabilitation, reinforcing intended movements through feedback and stimulation while recovery is still possible. As recovery plateaus, it shifts into an assistive role, driving wearable external devices for tasks like gripping objects. The company is also signaling longer-term ambitions in cognitive decline, Parkinson’s, and neuropsychiatric conditions.
The BCI landscape we’re tracking is moving fast. Just last month we covered how Neuracle’s minimally invasive device became the first commercially approved BCI in China, followed by increased funding activity:
StairMed raised $73M for its robot-inserted flexible electrode system;
Gestala raised $21M for a non-invasive ultrasound-based platform.
In the U.S., Merge Labs (backed by Sam Altman) raised $252M for ultrasound-based BCIs
Nia Therapeutics received FDA breakthrough device designation for an AI-guided memory loss BCI
Neuralink, the largest actor, plans high-volume production and a near-fully automated implantation workflow in 2026 following a $650M Series E.
We covered the broader neurotech landscape in our Neurotech Review here.
Read also:
Three Big Ideas in Aging Research That Could Shift the Therapeutic Landscape