Weekly Tech+Bio Highlights #2
ALSO: Success in One of the 2024’s Most Anticipated Clinical Trials; New AI-Docking Engine Goes Open-source; 2024 State of CRISPR Clinical Trials; AlfaFold 3 is Great, But Use With Care
Hi! I am Andrii Buvailo, and this is my weekly newsletter, ‘Where Tech Meets Bio,’ where I talk about technologies, breakthroughs, and great companies moving the biopharma industry forward.
If you've received it, then you either subscribed or someone forwarded it to you. If the latter is the case, subscribe by pressing this button:
Now, let’s get to this week’s topics!
News Highlights
🤝 Boehringer Ingelheim expands collaboration with OSE Immunotherapeutics to evaluate cancer drug prospects as cardiometabolic therapies, initiating a Phase 2 study and preclinical cancer immunotherapy program, with potential payouts reaching €1.1 billion.
🔬 GSK's long-acting asthma drug, depemokimab, shows success in Phase III trials, significantly reducing asthma attacks with a biannual dosing schedule.
📅 Bristol Myers Squibb anticipates an FDA approval decision for its subcutaneous Opdivo formulation by Dec. 29, covering all adult solid tumor indications.
✔️ FDA approves biosimilar versions of Regeneron’s Eylea, allowing interchangeable use, though patent litigation may delay market launch.
📉 Exscientia to cut 25% of workforce to save $40M annually, preserving its AI-driven drug pipeline, including key assets GTAEXS617 and EXS4318.
💰 Roche acquires U.S. gene-sequencing firm Genia Technologies for up to $350M, enhancing its gene sequencing capabilities with more accurate, faster, and less expensive technology.
🔬 Menten AI completes a research collaboration with Bristol Myers Squibb, using its generative AI platform to optimize peptide macrocycles.
⚙️ DeepCure introduces a high-throughput, multi-step robotic synthesis technology to address the limitations of manual chemical synthesis, potentially accelerating drug discovery and facilitating the creation and testing of complex small molecules, including AI-designed compounds.
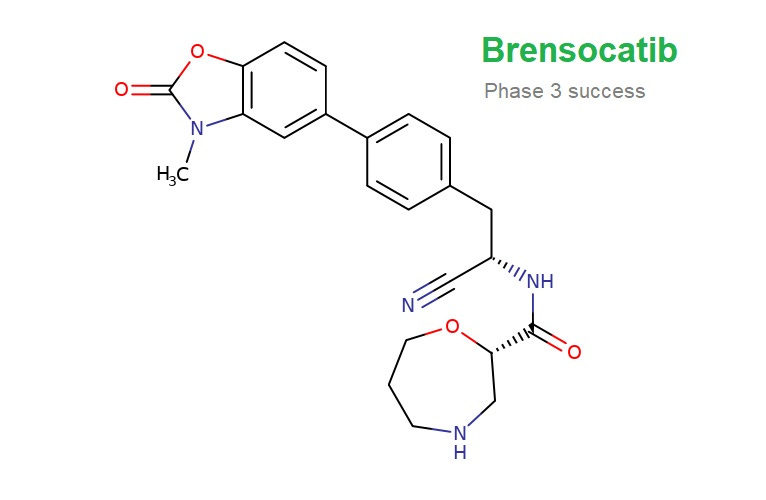
🔬 Insmed's Phase III ASPEN study reveals that brensocatib significantly reduces pulmonary exacerbations in non-cystic fibrosis bronchiectasis patients, meeting primary and secondary endpoints of reducing exacerbation frequency and improving lung function.
💰 ExpressionEdits raises $13M in seed funding, led by Octopus Ventures and redalpine, to advance its AI-based protein expression technology using intronization for optimizing gene design and protein production.
🔬 Turn Biotechnologies signs a global licensing agreement with HanAll Biopharma worth over $300 million to develop mRNA-based therapies for age-related eye and ear conditions, utilizing Turn Bio's Epigenetic Reprogramming of Age technology and eTurna delivery platform.
🔬 BenevolentAI and AstraZeneca advance heart failure treatment by incorporating a novel target discovered through BenevolentAI's AI-driven platform into AstraZeneca's discovery portfolio.
🔬 Enveda Biosciences partners with Microsoft to enhance its AI-driven natural product drug discovery, launching the PRISM model to "read and translate" mass spectrometry data into new medicines, leveraging Microsoft Azure for computational power.
🔬 Strand Therapeutics doses the first patient in a Phase I trial of STX-001, a programmable mRNA therapy targeting solid tumors, marking a milestone as the first such oncology therapy to enter clinical trials, aiming to improve immunotherapy efficacy with minimal toxicities.
🔬 Thousands of NHS cancer patients in England will participate in trials of personalized mRNA vaccines designed to prime the immune system to target and destroy remaining cancer cells, potentially reducing recurrence risks. The Cancer Vaccine Launch Pad has recruited 30 hospitals for this initiative, with initial treatments showing promise.
Success in One of the 2024’s Most Anticipated Clinical Trials
Experimental drug, brensocatib, from Insmed successfully reduced lung problems among patients with an airway disease in a closely watched Phase 3 trial, sending the company’s share price soaring.
The drug, brensocatib, reduced so-called pulmonary exacerbations by roughly 20% versus placebo in patients with bronchiectasis, hitting the trial’s primary endpoint.
The trial, called the ASPEN study, tested two dosages of the drug, and the company said both significantly cut rates of pulmonary exacerbations.
As part of the ASPEN study's conduct, more than 460 trial sites were engaged in nearly 40 countries, which later was adjusted to 391 sites and 35 countries.
The primary efficacy analysis included data from 1,680 adult patients and 41 adolescent patients.
Notably, Insmed is advancing an early-stage machine learning (ML)-supported research engine including protein engineering, gene therapy, and protein manufacturing.
I should say, the market clearly welcomed recent news by Insmed
A new AI-enabled Docking Engine Goes Open-source
Beijing-based DP Technology, a producer of scientific software, has open-sourced Uni-Mol Docking v2, an advanced AI-based molecular docking engine, designed to enhance the efficiency and accuracy of early-stage drug discovery by leveraging deep learning models.
Uni-Mol Docking v2 employs pre-trained molecular and pocket encoders to predict protein-ligand interactions with superior accuracy. This model achieved over 77% of ligand poses with RMSD values under 2.0 Å, surpassing traditional methods like Autodock Vina and addressing issues such as chirality inversions and steric clashes.
The model integrates deep learning to understand complex molecular representations, significantly reducing computational costs compared to traditional physics-based methods. Its performance has been benchmarked against AlphaFold3, demonstrating its capability in providing high-accuracy docking predictions.
Access Uni-Mol Docking v2
Ready-to-use Webapp: https://bohrium.dp.tech/apps/unimoldockingv2
Github: https://github.com/dptech-corp/Uni-Mol/tree/main/unimol_docking_v2
Paper: https://arxiv.org/abs/2405.11769
The State of CRISPR Clinical Trials in 2024
A pretty remarkable report by the Innovative Genomics Institute, highlighting the current state of CRISPR-based therapies.
This quote summarizes the current sentiment in the field:
“At this point, all hypotheticals, the words ‘potentially’ and ‘could’ or ‘in principle’ are gone,” says Fyodor Urnov, IGI’s Director of Technology & Translation. “CRISPR is curative. Two diseases down, 5,000 to go.”
To be fair, a lot of challenges remain to be overcome:
Technological Access: Delivering CRISPR-based therapies like Casgevy is technologically challenging and requires specialized facilities and expertise, limiting its availability to a few locations.
Financial Access: The high cost of CRISPR treatments, around $2 million per patient, presents financial access issues, especially in healthcare systems that may not cover such expensive therapies.
Financial Pressures on Companies: The high cost of clinical trials and the demand for quick returns on investment have led to layoffs and a narrowed focus on advanced products, rather than expanding new treatments.
Chemotherapy Risks: The pre-treatment chemotherapy required for CRISPR therapies is harsh on patients and carries significant risks, highlighting the need for safer conditioning methods.
Manufacturing Challenges: The complex and high-cost nature of manufacturing CRISPR therapies remains a bottleneck, affecting the scalability and wider adoption of these treatments.
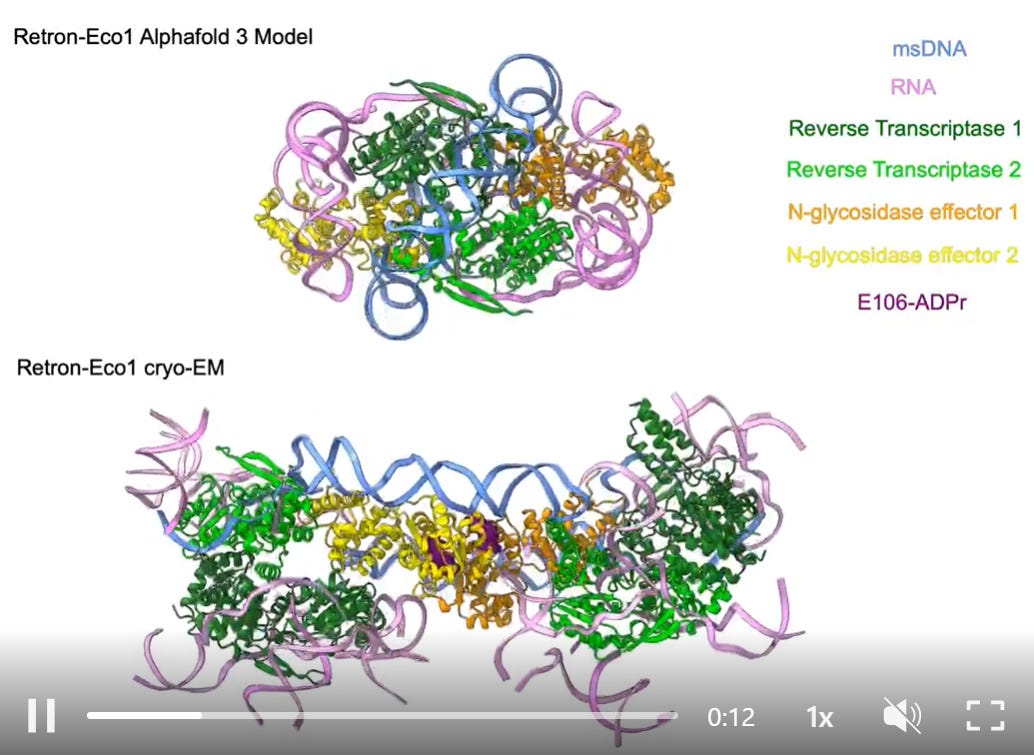
AlfaFold 3 Is a Monumental Achivement, But Use It With Care
With all the great possibilities AlfaFold 3 has brought us, we should remember that the quality of predictions varies significantly.
In this LinkedIn post, Guillermo Montoya, Professor & Research Director @ Novo Nordisk Foundation Center for Protein Research, compared AlfaFold 3 prediction with the actual Cryo-EM derived structure, and you can see for yourself, they are pretty different.
So, use AlfaFold 3 and enjoy great results, it is super useful, but do it with care.
You may also want to check our Thursday deepdive, dedicated to research marketplaces in the life sciences.
19 Life Science Marketplaces to Try in 2024
There I summarized 19 notable marketplaces categorised into the following areas of scientific need:
Life sciences marketplaces for various research products (consumables, reagents, research materials)
Life sciences roduct search & comparison engines
Life sciences R&D outsourcing services
Life sciences marketplaces for hiring talent
Biospecimens procurement and health data
Fine chemicals procurement (building blocks, screening compounds, intermediates)
Some Topical Newsletters You May Like:
AI Foundation Models in Biotech: New Paradigm
How Industry Embraces Organ-on-Chips: A 2024 Status Report
A Landscape of Novel Antibody-Drug Conjugates
11 Biopharma Trends to Watch in 2024
Using Quantum-Enhanced AI to Design Cancer Drugs For the First Time
It’s Been a Decade of AI in the Drug Discovery Race. What’s Next?