Simulating the Control Arm: Virtual Patients at the Trial Bottleneck
Digital twins promise smaller, faster trials, and the regulatory scaffolding is forming. But there’s still a validation gap.
Clinical trials remain the most expensive bottleneck in drug development. And although this stage comes after all the high tech pharmacological tinkering is over, a trial conduct runs into its own obstacles.
⚠️ The most immediate one is patient recruitment. Far back in 1979, the father of clinical pharmacology Louis Lasagna observed that the pool of eligible patients shrinks by 90% the moment a trial opens, only to reappear once it closes. Lasagna’s Law remains as relevant as ever: according to a 2022 article, 11% of trial sites enrol zero participants and nearly 90% of trials face meaningful delays. With Phase II and III trials costing roughly $40,000 per day, the financial toll is brutal.
⚠️ Another issue is clinical attrition. Research from VU Amsterdam found that between 2012 and 2019, the share of trials successfully completing each phase declined steadily—particularly at Phase II. In the first half of 2024, nearly a third (32%) of trials were discontinued at Phase II—a 56% rise compared to pre-pandemic levels. Combined with stagnant rates of Phase III initiation over that same decade, the picture is one of a systemic bottleneck: trials that begin are increasingly unlikely to see the finish line.
⚠️ Rare disease research presents its own distinct challenge. As the FDA’s Rare Disease Evidence Principles note, shrinking patient populations make it progressively harder to generate reliable efficacy data through conventional designs—especially placebo-controlled trials, where enrolling enough participants to reach statistical significance can be close to impossible.
⚠️ Apart from operational intricacies, there is the ethical dilemma. Randomized controlled trials remain the gold standard for evaluating new therapies, but randomization isn’t always defensible. When an effective treatment already exists, assigning patients to a placebo raises serious moral questions, e.g. HIV cure trials with the antiretroviral treatment interruption.
The question, then, is whether parts of the control process can be simulated rather than physically recruited.
Digital twins are emerging as a compelling response. Last October, Sanofi Ventures invested in a digital twin platform developer QuantHealth, bringing its total funding to $30M. In 2025, the FDA announced plans to phase out animal testing requirements for monoclonal antibodies in favor of human-relevant methods, including AI-driven computational models—with the EMA moving in the same direction. Both industry and regulators, it seems, are taking this technology seriously.
👥 How it Works
A digital twin is a virtual replica of a physical object, continuously updated with real-world data so it mirrors the original’s behavior in real time. The concept, first applied by NASA in the 1960s, has since migrated from engineering into healthcare.
The applications are wide-ranging: optimizing industrial processes as Eli Lilly did to boost production of their GLP-1 drugs, predicting equipment failures, streamlining supply chains, and accelerating product development.
In a clinical trial patients are generally divided into two groups, also known as arms. The intervention arm receives the experimental treatment; the control arm receives a placebo, standard-of-care treatment or sham, serving as the baseline against which results are measured. Randomized controlled trials (RCTs) are the gold standard because randomization minimizes bias, but that randomization isn’t always flawless.
The traditional workaround of external controls drawn from historical trials, health records, or registries all carry their own limitations. For instance, data like those don’t include underrepresented groups or don’t account for placebo effect due to their observational nature.
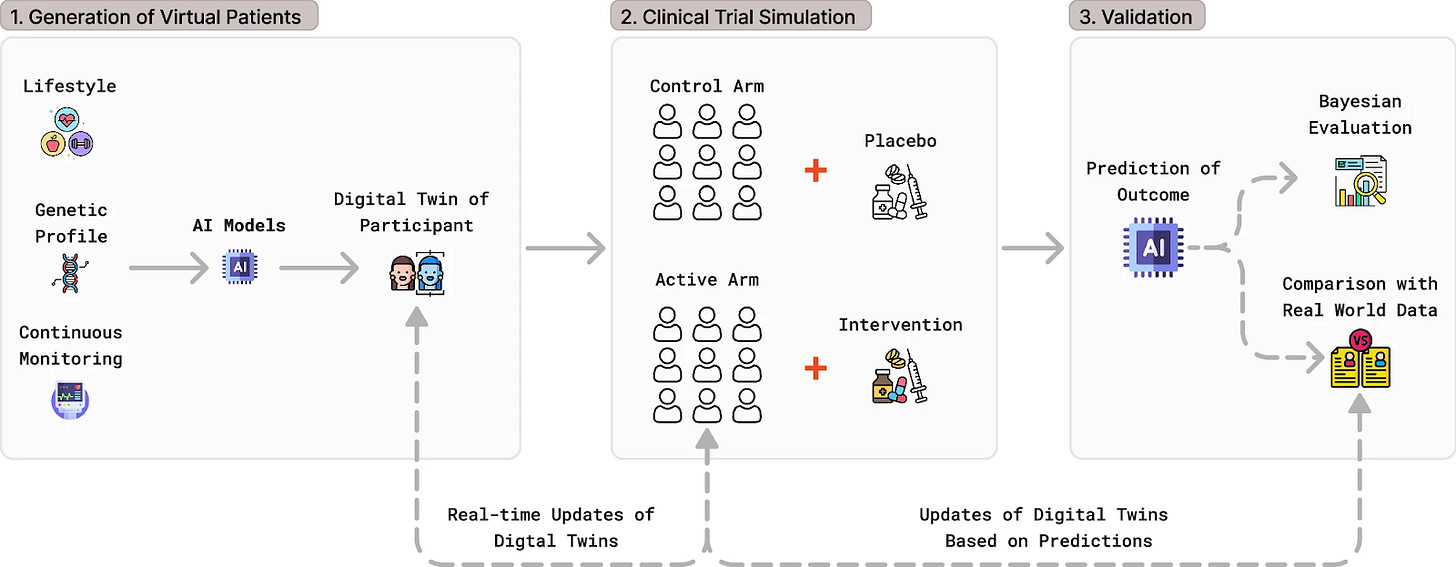
Digital twins go a step further: using AI models augmented with historical data, they generate individualized predictions of how a patient might respond under different treatment scenarios. When used to simulate outcomes for patients who do not receive the experimental therapy, these models can produce a synthetic control arm.
These trial-level twins build on a foundation of patient-specific digital twin modeling (virtual replicas of individual physiology shaped by genomics, imaging, and clinical history) which we covered in our earlier overview of biological and patient-specific twins.
In clinical trials, an AI-powered digital twin typically operates in three steps:
Build virtual patients — AI integrates biomarkers, imaging, genetics, and real-world evidence to generate synthetic profiles capturing the full variability of real populations.
Run simulated trials — virtual cohorts replace placebo groups or test experimental therapies in silico, probing efficacy and safety without exposing patients to unnecessary risk.
Optimize continuously — trial parameters like dosing and sample size are continuously refined in real time, anchored by validation against real-world data.
A less computationally demanding synthetic control arm approach uses AI-generated patient data based on registries, and real-world evidence but unlike DTs not modelling it on a particular individual. The appeal is sharpest in rare diseases, where finding enough eligible control patients is often impractical. The FDA, EMA, and NICE have all endorsed the approach, and it’s gaining traction: recent Phase II/III myeloma and lymphoma trials have leaned on external control data, and in at least one case (blinatumomab for acute lymphoblastic leukemia), a synthetic control arm helped support accelerated regulatory approval. AstraZeneca used over 300M synthetic patient records to advance its clinical trials, allegedly saving up to $100M per drug in development.
Synthetic control arms built from historical data have already supported label expansions and accelerated approvals with alectinib, blinatumomab, palbociclib among them. AI-generated individualized digital twins, however, have not yet served as primary evidence in a completed approval.