Companies on the Cutting-Edge of Antibiotic Discovery (Tech-enabled)
The looming health crisis; notoriously 'unwanted' business; recent (cool) breakthroughs in antibiotics; how tech may help revive the lost art of antibiotics discovery; NOT MEDICAL ADVICE.
Hi! I am Andrii Buvailo, and this is my weekly newsletter, ‘Where Tech Meets Bio,’ where I talk about technologies, breakthroughs, and great companies moving the industry forward.
If you've received it, then you either subscribed or someone forwarded it to you. If the latter is the case, subscribe by pressing this button:
Now, let’s get to this week’s topics!
Since the revolutionary discovery of penicillin in 1928 by Scottish bacteriologist and Nobel laureate Alexander Fleming, numerous inventions of new antibiotic classes have followed. A cascade of discoveries over decades pushed the limits of medicine in its ability to fight deadly infections—those that used to kill millions of people in the previous centuries.
A "silent pandemic"
According to the statistics from The Center of Disease Control and Prevention in the United States (the CDC), around 2.8 million people get infected by antibiotic-resistant illnesses in America each year, of whom around 35,000 die each year because antibiotics simply don’t work as they used to in the not-so-distant past. According to another source, drug-resistant diseases kill around 700,000 people each year globally, but a United Nations interagency group on antimicrobial resistance estimates that this could swell to 10 million a year by 2050 if no action is taken. This is more than the number of people who currently die from cancer worldwide every year.
We have been successfully using antibiotics in so many cases and for such a long time that we have started taking this powerful tool of modern medicine for granted. Meanwhile, the warning signs making headlines inform the public about emerging “superbugs” able to withstand antibiotics due to multidrug resistance and thereby becoming a deadly risk.
As widely used therapeutics such as tetracycline, erythromycin, and vancomycin lost much of their effectiveness against bacterial infections over the years, the antibiotics Colistin and Carbapenem are considered the big guns—a last line of defense when no other antibiotics are working, for example in the case of multidrug-resistant Gram-negative infections.
Such medications are called drugs of last resort, and they usually possess drastic side effects, as in the case of Colistin, which is toxic to the human kidney. Still, they are the last hope for some desperate patients. Now even this last weapon against malicious bacteria is becoming obsolete. As reported in recent years, mcr-1, a gene that confers resistance to Colistin, has been found in E. coli from over 30 countries, including cases of resistant bacteria isolated in China, the United States, and other regions.
The same story happens in the case of Carbapenem when the gene blaNDM-5 renders bacteria resistant to its action. In 2012, the CDC identified Carbapenem-resistant infections in about 4 percent of US hospitals. Here is recent research (March 2022) explaining the statistics and dynamics of the spread of the gene blaNDM-5 and its contribution to the rise of global antibiotic resistance.
The problem goes beyond treating infections. According to Army Col. Emil Lesho, director of the Defense Department’s Multidrug-resistant Organism Repository and Surveillance Network in the United States, the growth of bacterial resistance puts humanity at risk of losing access to modern medical “miracles”, such as medical surgeries, joint replacements, organ transplantation, cancer chemotherapies etc. These treatments can not be safely performed without antibiotics because it is almost impossible to avoid bacteria penetrating the body during severe medical interventions.
Reasons for the crisis?
The history of antibiotic research is the history of a constant race between drug discovery researchers and their ever-evolving natural enemy, bacterial infections. Since the discovery of early antibiotics, it has been noticed that bacteria evolve rather quickly, adjusting to the new environment and developing resistance to external molecules. Once resistance was developed, rendering the existing antibacterial drug useless, a new type of antibiotic was needed to save the day.
The drug discovery industry used to do a good job of keeping up with natural bacterial threats, and numerous new classes of antibiotics were discovered in the middle of the last century. However, the progress slowed down and almost vanished by the 2000s. In fact, researchers haven’t identified a new class of antibiotic medication since 1987, while bacteria kept developing resistance to the previously invented drugs. So why has innovation slowed so dramatically?
A major reason for the substantial decline in novel antibiotic approvals in the past three decades is the economics of drug development for antimicrobials. Pharmaceutical companies spend hundreds of millions of dollars on drug discovery research, multi-step clinical trials, and the FDA approval process; thus, the result has to pay back very well, for a long time, and with minimum business risks.
It stimulates companies to concentrate on therapeutic areas where there are chances to develop blockbuster drugs. An ideal candidate would be demanded by millions of people on a regular basis for a very long period of time, like pills to keep cholesterol in check, normalize blood pressure, or maintain sexual activity. Another very crowded space is oncology research and drug discovery.
But antibiotics are among the worst candidates, businesswise (at least, until recently).
They have strict prescription limits and should only be used for about a week or longer. They are relatively cheap, for example, compared to anti-cancer drugs, and worst of all, they compete in a field of inexpensive generics making it hard to gain profits. On top of that, antibiotics can become obsolete at any time due to the development of bacterial resistance, and this is a substantial risk for businesses.

Furthermore, according to a World Health Organization report released in 2021, none of the 43 antibiotics currently in development "adequately address the problem of drug resistance" in the bacteria thought to be the most harmful to public health.
The lost (and reviving) art of antibiotic discovery
However, besides purely business-related reasons, there are other reasons why we have not seen major innovations in the antibiotic space ever since the 'golden age' of antibiotic discovery. Such reasons are brilliantly summarized in a recent paper: "Brief Overview of Approaches and Challenges in New Antibiotic Development: A Focus On Drug Repurposing", such as the collapse of the Waksman Platform and shift from soil-based discovery to other strategies (which proved to be largely unsuccessful). It was also a shift from phenotypic discovery of antibiotics (in vitro growth inhibition assays) to target-based discovery and HTS, which was all widely regarded as a progressive move (and it proved so in many therapeutic areas), but proved to be a failure specifically in the antibiotics space. An increasing focus on medicinal chemistry efforts in contrast to reliance on natural sources of inspiration and an industry bias towards Rule-of-5 molecules were also possible contributors to the overall decreasing productivity in terms of being able to discover new classes of antibiotics.
According to Dr. Chris De Savi, VP of Drug Discovery at Kymera Therapeutics, only 17 new systemic antibiotics and 1 related biologic have been approved by the FDA since 2010. Among these drugs, 14 were approved for common bacterial infections; 1 was approved for Clostridioides difficile infection (CDI); 1 was licensed to prevent CDI recurrence; and 2 were approved for drug-resistant tuberculosis. Unfortunately, the majority of these new drugs represent modifications to existing chemical structures rather than new drug classes.
"In contrast to the Gram-positive field, where a variety of treatment options are available, the resistance situation in infections caused by Gram-negative bacteria is dramatic. Pan-resistant Gram-negative enterobacteriaceae or non-fermenters have only increased lately." Dr. Chris De Savi further notes.
Reviving the field of antibiotic discovery
No matter the reasons of the antbiotics discovery crisis over the last several decades, the situation with the growing threat of antibiotics resistance keeps worsening, and the industry, as well as governments, are trying to change the "anti-trend" towards more productive realms.
In 2012, the United States administration introduced the Generating Antibiotics Initiatives Now (GAIN) Act, which was designed to provide incentives for pharma businesses and investors to play the game of antibiotics discovery. The Act extended the exclusivity of new antibiotics by five years over any existing exclusivity, such as the ongoing patent protection, Hatch-Waxman, orphan drug or pediatric exclusivity. It gave extra-time for pharmaceutical companies to market antibiotics without the need to compete with generics. In addition, all new antibiotics that fall under the GAIN provisions receive fast track and priority review status which substantially accelerates the approval process with FDA.
The recently introduced Pioneering Antimicrobial Subscriptions to End Upsurging Resistance (PASTEUR) Act would establish a subscription-style payment model whereby the federal government would pay upfront for access to FDA-approved antibiotics that target drug-resistant pathogens and address urgent, unmet medical needs. The purpose of the bill, which would decouple company profits from the amount of antibiotics sold, is to assist in resolving market issues that have caused many pharmaceutical companies to stop developing antibiotics and contributed to the lack of new, innovative antibiotics in the pipeline.
A new wave of players and innovations
While antibiotic drug discovery remains a notoriously difficult area for entrepreneurship, new players emerge in this area every year. In 2020, the Boston-based AMR Action Fund, supported by pharmaceutical companies and organizations like the Wellcome Trust, began funding early-stage research and clinical trials of antimicrobial drugs at smaller biotechnology companies. The fund dedicated $1 billion to invest in antibiotics companies and has several antibiotics companies in its portfolio, including Bioversys, a clinical-stage Swiss pharmaceutical company focusing on research and development of small molecules acting on novel bacterial targets with applications in antimicrobial resistance (AMR) and targeted microbiome modulation. The company's pipeline addresses nosocomial infections of Acinetobacter baumannii (BV100, Phase 2 ready) and tuberculosis (BVL-GSK098, Phase 2 ready) in collaboration with GSK and a consortium of the University of Lille.
AMR Action Fund's portfolio also includes Venatorx Pharmaceuticals is a private company that is focused on the discovery and development of novel anti-infectives to treat multi-drug-resistant (MDR) bacterial infections and hard-to-treat viral infections. Both of Venatorx's lead antibacterial clinical-stage programs, cefepime-taniborbactam and ceftibuten/VNRX-7145, are combinations of broad-spectrum beta-lactam and beta-lactamase inhibitors. Additionally, Phase 1 clinical development has begun on the first Venatorx antiviral compound, VNRX-9945, an inhibitor of the Hepatitis B virus. Penicillin-binding protein (PBP) inhibitors are a novel class of non-beta-lactam antibiotics that the company is developing as part of its discovery-stage programs. These inhibitors have the potential to overcome the 70+ years of resistance to penicillin and usher in a new era of antibacterial therapeutics.
Founded in 2013 and traded publicly on Nasdaq since 2017, Spero Therapeutics is a multi-asset, clinical-stage biopharmaceutical company focused on identifying, developing, and commercializing novel treatments for bacterial infections, including multi-drug-resistant, or MDR, bacterial infections and rare diseases. SPR720 is an oral antimicrobial agent in development by Spero Therapeutics for the treatment of nontuberculous mycobacterial (NTM) pulmonary disease, a rare orphan disease. The IV administration of SPR206 is being researched as a potential new way to treat multidrug-resistant Gram-negative bacterial infections in hospitals. To treat cUTIs, including pyelonephritis, caused by specific microorganisms in adult patients with few other oral treatment options, tebipenem HBr was developed as the first broad-spectrum oral carbapenem-class antibiotic.
Founded in 2015, Entasis Therapeutics, a spinout of AstraZeneca, raised almost $200 million in multiple rounds and managed to build a robust pipeline consisting of clinical and pre-clinical pathogen-targeted small-molecule antibacterials for the treatment of multidrug-resistant Gram-negative bacteria.Entasis's drug discovery platform combines the use of genetic tools, molecular dynamics simulations, and modeling to enable the exploration of novel therapies in a highly directed, focused manner.
Discovery of Teixobactin and Clovibactin using iChip
In 2015, a group of scientists led by Dr. Kim Lewis, Director of the Antimicrobial Discovery Center at Northeastern University, reported a new antibiotic, teixobactin, capable of killing several types of bacteria, including antibiotic-resistant strains of tuberculosis and staphylococcus (MRSA infections), without detectable resistance developing over time. Teixobactin is a small-molecule antibiotic of a new class capable of destroying 'drug-resistant' bacteria.
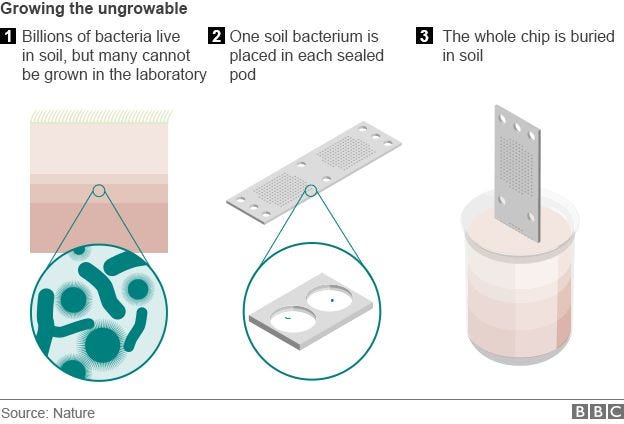
What is even more groundbreaking is the way in which the compound has been found. Researchers developed an innovative reinvention of an old technique used in the past for many of the antibiotic discoveries of the mid-twentieth century: soil samples.
Combing soil samples for microbes producing their own antibiotic compounds to kill competing bacteria was once a powerful technique by which many of the early antibiotics were found. However, when all the compounds that were easiest to find this way and cultivate in a laboratory were identified, innovations largely dried up.
Dr. Lewis and his colleague at Northeastern, Dr. Slava Epstein, revived the soil mining approach by inventing the iChip. This device allows the cultivation in a lab of bacteria that were impossible to cultivate by previous techniques. In this new approach, soil samples are placed between the iChip membranes, and the device is buried back in the ground where bacteria can get nutrients from the soil in a natural way. Once colonies grow within the iChip, they are transferred back to the lab to discover antibiotics.
Overall, about 50 000 strains of uncultured bacteria have been grown using the iChip, and 25 new promising antibiotic compounds have been identified, including teixobactin. The new invention has the potential to solve the biggest issue in antibiotic research: the lack of drug candidates. Systems like the iChip enable the exploration of large numbers of compounds with promising antibacterial properties, which is the key to fighting the resistance phenomenon.
Northeastern University licensed the patent on the iChip technology and any compounds produced to an early-stage biotech company, NovoBiotic Pharmaceuticals (Cambridge, MA), founded by the researchers. The research is currently ongoing to follow up on the teixobactin discovery and translate it into clinical practice. For instance, in August 2022, a major Nature article was published in which an international coalition of laboratories, including NovoBiotic, reported that teixobactin kills bacteria through a two-pronged attack on the cell envelope.
Recently, a study published in Cell identified a potent new antibiotic named Clovibactin from soil bacteria previously thought to be "unculturable".
Most of the antibiotics we use today, like streptomycin, vancomycin, and tetracycline, come from bacteria in the soil. But here's the thing: we can't grow about 99% of these bacteria in labs. So, we're missing out on studying them for potential new antibiotics.
This research, led by Utrecht University in collaboration with NovoBiotic Pharmaceuticals and Northeastern University, leverages iChip, enabling the growth of bacteria in their natural environments. The antibiotic was isolated from a species named Eleftheria terrae ssp. Carolina, found in North Carolina's sandy soil. Clovibactin's unique mechanism binds to three vital precursor molecules crucial for cell wall construction, making it challenging for bacteria to develop resistance.
When tested on mice, clovibactin efficiently combated Gram-positive bacterial infections resistant to other antibiotics, comparable in efficacy to the well-known antibiotic vancomycin.
Discovery of Darobactin from animal microbiome
In 2019, a team of researchers led by Dr. Kim Lewis announced their discovery of Darobactin, which can kill resistant gram-negative bacteria. The substance was discovered by Yu Imai, a postdoctoral research associate in Lewis' lab, from Photorhabdus bacteria that reside inside the gut of a nematode, a tiny parasitic worm found in soil. Lewis claims that this is the first time an antibiotic with potential for human use has been discovered in the animal microbiome.
Darobactin measures 965 Daltons, which is unusually large for an antibiotic. That made it unclear how darobactin functions since it is too large to pass through gram-negative bacterial membranes. The scientists repeatedly exposed E. coli to the substance until bacterial resistance developed in order to find out. These bacteria had mutations in a gene that produces the BamA molecule, which covers the bacteria's outer membrane. It was a very interesting finding—an outer-membrane protein.
According to Karen Bush, a biochemist at Indiana University, not all gram-negative bacteria are inhibited by the substance at low concentrations. According to her, Darobactin may serve as a novel scaffold that drug chemists can modify to create a more widely effective medication.
Discovery of Dynobactin via computational screening
Another recent success aimed at tackling Gram-negative bacteria was the discovery of Dynobactin in 2022. It is a new antibiotic that was discovered and its mechanism of action identified by an international team of researchers at the University of Basel in Switzerland. The study was conducted by the researchers in collaboration with Professor Sebastian Hiller from the University of Basel's Biozentrum as part of the NCCR "AntiResist" initiative, with results published in Nature Microbiology.
The fact that many bacteria produce antibiotic peptides to compete with one another was exploited by the researchers. Further to that, unlike natural substances, these peptides are encoded in the bacterial genome. It was found that the genes for such peptide antibiotics share a characteristic feature, and using this feature, the computer thoroughly screened every molecule in the genome of the bacteria that produce these peptides and eventually landed on the discovery of Dynobactin.
The bacterial membrane protein BamA, which is crucial for the development and upkeep of the outer-protective bacterial envelope, is blocked by this peptide. Despite having very few chemical similarities to the previously identified Darobactin, Dynobactin nonetheless targets the same area of the bacterial surface, which was an unexpected discovery.
AI-driven discovery of Abaucin
In May 2023, Scientists from McMaster University and the Massachusetts Institute of Technology utilized artificial intelligence to discover a new antibiotic effective against the superbug Acinetobacter baumannii, identified by the World Health Organization as a critical health threat. This superbug can survive on surfaces for extended durations and can result in various infections, including in the urinary tract and lungs. The AI algorithm screened thousands of antibacterial molecules, pinpointing the newly identified antibacterial compound named abaucin. After using the trained AI model to analyze over 6,000 compounds, lab testing confirmed nine potential antibiotics, including abaucin.
LPC-233 targets gram-negative bacteria
Recently, Duke University researchers annouced a synthetic molecule, LPC-233, that combats gram-negative bacteria like Salmonella and E. coli by interfering with the bacterium's ability to form its outer lipid layer. The molecule was effective against all 285 bacterial strains it was tested on, including those highly resistant to existing antibiotics.
The compound is credited to the dedication of late Duke Biochemistry Chair Christian Raetz, and its target is an essential enzyme for bacterial outer membrane creation.
Pei Zhou, the lead investigator, and his colleague, Eric Toone, have co-founded Valanbio Therapeutics to progress LPC-233 through clinical trials. The research, supported by various grants, was recently published in Science Translational Medicine.
Afterthought
We have reviewed some cool recent breakthroughs, including AI-generated drugs and antibiotics derived via the iChip platform. But we shall remember that those discoveries are still years away from being available for prescription (if ever). Currently, the situation with growing antibiotic resistance is alarmingly difficult and is worsening, still.





Insightful article, Andrii!
"according to a World Health Organization report released in 2021, none of the 43 antibiotics currently in development "adequately address the problem of drug resistance"" Wow 😬