How Technology Enables Better Clinical Trials
On artificial intelligence, precision biomarkers, better clinical trials, and a decentralization trend.
Hi! I am Andrii Buvailo, and this is my weekly newsletter, ‘Where Tech Meets Bio,’ where I talk about technologies, breakthroughs, and great companies moving the industry forward.
If you've received it, then you either subscribed or someone forwarded it to you. If the latter is the case, subscribe by pressing this button:
Now, let’s get to this week’s topics!
Disclaimer: This post is not medical advice of any kind, it is for informational and educational purposes only.
How AI Empowers Biomarker-Driven Clinical Trials
(This article is sponsored by Lantern Pharma (NASDAQ: LTRN))
Clinical trials are the lifeline of modern medicine, bridging the gap between lab-borne scientific discoveries and the availability of life-saving treatments for patients. Unfortunately, the success rate of clinical trials, particularly in the field of oncology, is not as robust as one might hope. In fact, a 2019 study of 7,455 interventional phase trials in oncology conducted from 2000 to 2015 suggests an estimate that only 3.4% of cancer drugs that enter Phase I clinical trials ultimately receive FDA approval. Many of these failures can be attributed to obstacles such as poor trial design and inefficient patient selection, often resulting from a lack of biomarker-driven insights. As a result, drug development can be a laborious and expensive process, with the cost of bringing a new drug to market estimated to be around $2.6 billion. As we grapple with these challenges, there is an urgent call for more efficient and targeted methods of drug development, and the answer may lie in the utilization of novel actionable biomarkers and the power of artificial intelligence (AI) to plan and execute clinical trials. For instance, IQVIA's artificial intelligence-based platform, using their Real World Data assets, have enabled precise patient and healthcare professional (HCP) targeting, doubling identified eligible patients, finding 30% more with uncontrolled symptoms, predicting 81% likely to quit treatment early, pinpointing events linked to early discontinuation, and boosting treatment transition success by 500% compared to prior methods.
What is a clinical trial?
Clinical trials form an integral part of the drug development process. They are carefully designed studies involving human participants, undertaken to determine the safety and efficacy of a new drug or medical device. This process is usually divided into four stages. Phase I trials primarily assess safety and dosage in a small group of healthy volunteers or patients. Phase II trials involve a larger group of patients and aim to assess the drug's efficacy and side effects. Phase III trials test the drug in an even larger group of patients over a longer period of time to confirm its effectiveness, monitor side effects, and compare it to commonly used treatments.
Designing clinical trials for central nervous system (CNS) drug evaluation, for example, for brain tumors, represents an additional challenge due to unique properties of the blood-brain barrier (BBB). The BBB's unique and stringent selectivity often necessitates the implementation of specialized Phase 0 trials in CNS oncology programs. Because of BBB, these trials require higher systemic drug concentrations to achieve detectable levels within the CNS, as compared to other therapeutic areas, increasing the complexity and risk of the process.
The arena of clinical studies encompasses various approaches, including double-blind studies and more transparent strategies like open-label trials. With high failure rates in drug development, biomarkers are emerging as pivotal components of clinical trial experiment design, refining patient selection for more accurate outcomes. The trend towards using master protocols indicates a push for efficiency, as this model allows simultaneous examination of various treatments. More specialized frameworks, like basket and umbrella trials, further incorporate factors such as genetic biomarkers, reflecting the industry's shift towards more nuanced methodologies.
The Evolution of Clinical Biomarkers
Advancements in Next-Generation Sequencing (NGS) and our understanding of the human genome have revolutionized the field of biomarker discovery, particularly in oncology. Traditional biomarkers often utilized circulating markers in blood, plasma, and serum, or those detectable through imaging techniques. However, with the advent of genomics, we have been able to delve deeper, identifying individual genetic variations that play pivotal roles in disease pathology, specifically in cancers where genetic mutations often take center stage.
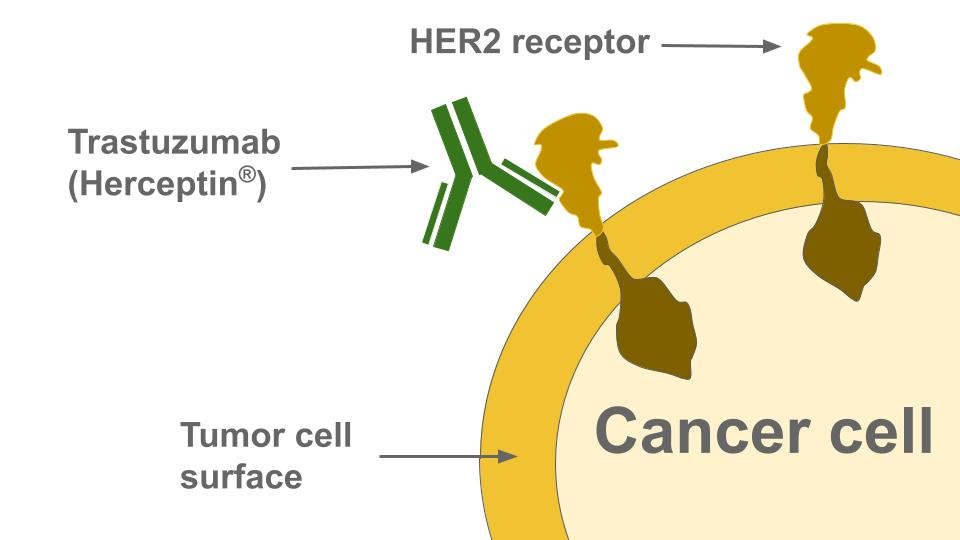
One of the defining moments in this biomarker-led therapeutic revolution was the approval of Herceptin (trastuzumab), a monoclonal antibody, by the FDA in 1998. Designed to target the HER2 protein, it was used to treat HER2-positive metastatic breast cancer, either as a first-line therapy in combination with other drugs, or as a standalone second-line treatment. This represented a significant stride forward in targeted cancer therapy, and opened the floodgates for similar advances. Today, we have a plethora of HER2-targeting drugs, including Tyrosine Kinase Inhibitors (TKIs) such as neratinib, lapatinib, and tucatinib, which have broadened the therapeutic scope beyond breast cancer to other cancers that overexpress the HER2 protein or exhibit HER2 gene amplification.
HER2's sibling in the human epidermal growth factor receptor (ErbB) family, EGFR (also known as HER1), has been another game changer in the oncology biomarker landscape. EGFR mutations or overexpression are commonly found in many cancers, including glioblastoma, lung cancers, colorectal cancer, head and neck cancer, and breast cancer. The development of EGFR-inhibiting drugs such as TKIs (erlotinib, gefitinib, lapatinib) and monoclonal antibodies (cetuximab and panitumumab) have further emphasized the significant role of biomarkers in targeted cancer therapy.
Biomarker-driven clinical trials
The story of biomarker evolution in oncology doesn't stop here. Apart from HER2 and EGFR, numerous other biomarkers like KRAS, BRCA1/2, and PD1/PD-L1 have shaped the therapeutic strategies for several cancers, including breast cancer, non-small cell lung cancer, and ovarian cancer. Additionally, an astounding number of 76 biomarkers have been identified as potential indicators of tumor invasiveness, including S100 proteins, annexins, galectins, CATD, TGM2, gelatinases, fibronectins, proteoglycans, TGFBI, FGG, APMAP, and more. The oncology biomarker discoveries underpin a remarkable shift towards personalized medicine, where treatment decisions are increasingly driven by the individual patient's unique genetic makeup.
In the intricate world of clinical trials, biomarkers serve as a guiding light, enabling more accurate prediction of therapeutic success and shaping the course of personalized medicine. Their contribution to medical progress has been highlighted by a comprehensive study that scrutinized over 10,000 clinical trials and focused on 745 drugs. The findings are powerful: introducing the factor of biomarker status significantly improved the prediction model for the trajectory of drugs throughout different clinical trial stages.
The 2021 study published in Cancer Medicine, employed two Markov models, which represent a statistical method that calculates the probability of transitioning from one state to another. When observing the effects of biomarker application across various types of cancers, the advantage was clear. The hazard ratios – the probability of a drug's approval – were significantly elevated when biomarkers were employed. For all indications combined, there was nearly a fivefold increase in the chances of drug approval. The most striking improvements were seen in specific cancers: a 12-fold increase for breast cancer, an 8-fold boost for melanoma, and a 7-fold growth for non-small cell lung cancer (NSCLC). Even for exploratory biomarkers that are not yet FDA approved, the model incorporating these biomarkers outperformed the model without biomarkers by 4-fold.
Nevertheless, biomarkers are not a foolproof guarantee of success. If they are not fully validated, they can potentially contribute to unsuccessful clinical trial outcomes.
Artificial Intelligence: A game changer in clinical trials
In the modern era, Artificial Intelligence, or AI, has found utility across a diverse range of sectors, including healthcare. AI refers to the development of computer systems capable of performing tasks that typically require human intelligence. It encompasses a range of applications, from learning from experience and interpreting complex data to drawing conclusions and making decisions.
Now, the world of clinical research, particularly biomarker-led trials, is witnessing an AI revolution. The sheer complexity and volume of data in these trials call for a technological intervention. AI, with its capacity to process vast amounts of data, detect potential biomarkers, and even forecast patient responses, is transforming how clinical trials are designed and executed. Already now, AI-powered tools demonstrate tangible improvements in oncology clinical trials, such as a 50% increase in identifying potentially eligible patients and a 25% reduction in time for patient screening. Moreover, in automating trial matching and eligibility determinations, AI algorithms, applied to both structured and unstructured data, have achieved accuracies up to 87.6% with agreements between manual professionals and AI algorithms ranging from 81% to 94% in areas like breast cancer trials.
AI-based platforms are becoming transformative tools in the clinical trials landscape. Notable companies like Lantern Pharma, Owkin, and Biotx.ai, to name a few, are leveraging AI platforms for improving drug discovery and clinical trials, marking strides in the industry.
To illustrate this, Dallas-based clinical-stage precision oncology company Lantern Pharma, for instance, developed AI drug discovery platform RADR® which leverages transcriptomic, genomic, and drug sensitivity data from diverse sources, analyzing a vast volume of over 25 billion clinical data points, 154 drug-tumor interactions, and 130,000+ patient records across 17 databases. By integrating public resources, commercial studies, and Lantern's ex vivo 3D tumor models, the platform establishes correlations between genetic biomarkers and drug activity. Through this comprehensive approach, RADR® enables the transformation of multi-omics data into predictive models, facilitating the identification of candidate biomarkers and enhancing patient stratification for optimized clinical trial design.
Utilizing their RADR® platform, Lantern Pharma tapped into AI to uncover the crucial role of the biomarker PTGR1 for their drug candidate LP-184. With this AI-led guidance, it became clear that LP-184 could be particularly effective against cancers with DNA Damage Repair (DDR) vulnerabilities. This insightful revelation, rooted in artificial intelligence, has paved the way for LP-184's upcoming clinical trials, aiming to target specific cancers such as pancreatic, prostate, ovarian, and breast that exhibit HR/NER pathway discrepancies.
In another example, Paris and New York-based precision oncology company Owkin developed AI-driven biomarker models to predict treatment response to immune checkpoint inhibitor (ICI) and prognosis in Non-Small Cell Lung Cancer (NSCLC) patients from clinical and histological data.
Owkin collaborates with leading academic centers to curate deep, multimodal data that is AI-ready, and they are developing a technology platform, Abstra, designed to expedite AI biomedical research by helping researchers discover collaborators and datasets. Through the application of AI to this rich data, they subtype patients, pinpoint novel biomarkers, and enhance drug discovery while reducing clinical trial risks.
Berlin-based Biotx.ai utilizes a combination of expert judgment, mechanistic validation, and artificial intelligence to identify drug candidates poised for success in phase IIb. Led by Jack Scannell, the expert drug selection committee assesses treatments using their synthetic clinical trial platform, which predicts drug efficacy and side effects based on independent mechanistic models. These potential treatments are unearthed using unique wide data algorithms that uncover intricate relationships between genes, revealing links between drug targets and diseases. Instead of the common big data approach, Biotx.ai focuses on wide data, deciphering complex patterns from limited examples by employing knowledge graphs to simplify hypothesis spaces, using predictive power algorithms. Their prowess was evident when they identified a link between variants on the CDK6 gene and severe COVID-19 outcomes, which was subsequently verified by the University of Bristol through in-vitro testing.
7th Annual Cell & Gene Therapy Innovation Leaders Summit 2024
Event Date: Feb. 7, 2024 - Feb. 8, 2024
Location: Spain, Barcelona, Atzavara Hotel & Spa
Website: 7th annual Cell & Gene Therapy Innovation Summit #CGTI 2024
Promo Code exclusive to our readers: BIOTREND20
With more regenerative medicine products approaching clinical testing and commercialisation, seize this opportunity to hear detailed insights and industry best practices from renowned presenters.
Achieve a deeper understanding of the many manufacturing and industrialisation burdens unique to carrying durable and curative therapies to needy patients.
The Shift to Decentralized Clinical Trials
As the FDA increasingly embraces decentralized clinical trials (DCTs), the industry is responding by exploring new ways to conduct clinical research. Decentralized clinical trials companies are leveraging technology such as telemedicine, mobile apps, and wearables to facilitate remote data collection and monitoring. This shift is driven by the need to improve trial efficiency, reduce costs, and enhance patient accessibility.
Pros of Decentralized Clinical Trials
Improved patient access and diversity: DCTs allow patients from various geographic locations and backgrounds to participate, leading to more representative and inclusive trials.
Reduced burden on patients: Patients can participate from the comfort of their own homes, eliminating the need for frequent travel to clinical trial sites.
Faster enrollment and retention: The convenience and flexibility of DCTs can lead to faster patient recruitment and reduced dropout rates.
Real-time data collection: Continuous monitoring through digital devices enables real-time data collection and analysis, improving the overall quality of data.
Cons of Decentralized Clinical Trials
Data security and privacy concerns: Protecting sensitive patient information and maintaining data integrity is critical, necessitating robust security measures.
Technological barriers: Limited access to technology or connectivity issues can exclude certain patient populations or introduce bias in the results.
Regulatory challenges: As DCTs are relatively new, regulatory bodies like the FDA are still developing guidelines to ensure compliance and safety.
Several companies have already adopted decentralized clinical trials with notable success. Science 37, a prominent DCT company, has collaborated with major pharmaceutical firms such as Novartis and Sanofi to conduct remote trials. In one particular case, their partnership with AOBiome led to the successful completion of a Phase 2b trial for a novel acne treatment, with 372 participants recruited in just three months.
Another example is Medable, which has developed a digital platform to streamline the DCT process. Their collaboration with PPD, a global contract research organization, has enabled more efficient and patient-centric trials across various therapeutic areas.
Several drug candidates have been tested or are being tested using DCTs. For instance, Pfizer's COVID-19 vaccine trials included a decentralized component, utilizing telemedicine and remote monitoring to manage a large participant pool.
RELATED: 9 Notable AI Companies in Clinical Research to Watch in 2023
Notable decentralized clinical trial companies:
Science 37 (https://www.science37.com/):
Science 37 is a leader in decentralized clinical trials, offering an end-to-end solution through its NORA (Network Oriented Research Assistant) platform. The platform includes features such as telemedicine, eConsent, ePRO, and eCOA, enabling remote patient participation and data collection for various therapeutic areas.
Medable (https://www.medable.com/):
Medable provides a flexible, modular platform for decentralized clinical trials, offering capabilities such as remote data collection, patient monitoring, and eConsent. Their platform aims to streamline the clinical trial process, increase patient engagement, and reduce study timelines.
THREAD Research (https://www.threadresearch.com/):
THREAD specializes in decentralized clinical trials with a comprehensive platform that includes telehealth, eConsent, ePRO, and eCOA solutions. They focus on offering patient-centric experiences, which can improve recruitment and retention rates in clinical trials.
PRA Health Sciences (acquired by ICON) (https://www.prahs.com/):
PRA Health Sciences (now ICON) offers a range of services for decentralized clinical trials, including remote patient monitoring, virtual site visits, and digital health tools. Their solutions are designed to provide flexibility and scalability, enabling sponsors to conduct trials efficiently and with improved patient outcomes.
Covance (https://www.covance.com/):
As a part of Labcorp, Covance provides services for decentralized clinical trials that include patient recruitment, remote monitoring, and digital endpoints. They leverage their extensive clinical research experience to offer tailored solutions for various therapeutic areas and study types.
IQVIA (https://www.iqvia.com/):
IQVIA is a global provider of advanced analytics, technology solutions, and contract research services. They offer a suite of solutions for decentralized clinical trials, including data management, virtual site visits, and patient engagement tools, designed to optimize trial efficiency and improve patient experiences.
Parexel (https://www.parexel.com/):
Parexel is a global clinical research organization that offers a variety of services for decentralized clinical trials. Their solutions include eClinical platforms, digital health tools, and remote patient monitoring, which aim to enhance patient engagement and streamline the clinical trial process.
Oracle Health Sciences (https://www.oracle.com/industries/life-sciences/):
Oracle offers a cloud-based platform for decentralized clinical trials, featuring data management, patient engagement, and remote monitoring solutions. Their integrated platform is designed to support innovative trial designs and improve trial efficiency.
Evidation Health (https://evidation.com/):
Evidation Health focuses on generating real-world evidence from decentralized clinical trials by leveraging digital technology for data collection and analysis. Their platform enables the measurement of patient behavior and outcomes outside of traditional clinical settings, providing valuable insights for healthcare decision-making.
ObvioHealth (https://www.obviohealth.com/):
ObvioHealth offers an end-to-end solution for decentralized clinical trials, including patient recruitment, remote data collection, and virtual site management. Their platform, ClaimIt, combines mobile technology, telemedicine, and data analytics to facilitate efficient and patient-centric clinical trials.
AI Technologies and Decentralized Clinical Trials
AI technologies are transforming various aspects of decentralized clinical trials, from patient recruitment to data analysis. Here are some key areas where AI is making a significant impact:
Patient recruitment and retention: AI algorithms can analyze vast amounts of patient data from electronic health records (EHRs), social media, and other sources to identify potential trial participants. This targeted approach improves recruitment rates and ensures a diverse and representative study population. Additionally, AI-driven chatbots and virtual assistants can improve patient engagement, leading to better retention rates throughout the trial.
Trial design optimization: AI can help optimize trial design by analyzing historical data and simulating different scenarios to predict outcomes. This can lead to more efficient study designs, reducing the duration and cost of trials, and enhancing the overall success rate.
Real-time data analysis: AI-driven analytics platforms can process and analyze large volumes of data collected during decentralized clinical trials, including data from wearables, mobile apps, and telemedicine consultations. This enables real-time monitoring, early detection of potential safety issues, and quicker decision-making.
Personalized interventions: AI technologies can analyze individual patient data to recommend tailored interventions, enhancing the patient experience and improving outcomes. In decentralized clinical trials, personalized medicine can lead to better treatment adherence and more accurate evaluations of drug candidates.
Regulatory compliance: AI can help decentralized clinical trials companies ensure compliance with FDA and other regulatory guidelines by automating data validation, monitoring data integrity, and identifying potential discrepancies in real-time.
Below let’s review eight companies making waves in the DCT landscape, showcasing the remarkable progress they've made in recent years. From blockchain-powered data management to AI-driven patient recruitment, these cutting-edge organizations are breaking down traditional barriers and empowering patients to participate in trials with greater ease, flexibility, and convenience.
The space is dominated by large contract research players, like IQVIA, In this post, we will review eight relatively young companies enabling decentralized clinical trials.
Deep 6 AI
Deep 6 AI is a health technology company founded in 2015, specializing in artificial intelligence (AI) solutions for decentralized clinical trials. Their platform focuses on patient recruitment, trial feasibility, and site selection by analyzing structured and unstructured real-world data from electronic health records (EHRs) and other sources.
Using natural language processing (NLP) and machine learning algorithms, Deep 6 AI's platform identifies eligible patients for clinical trials based on specific criteria such as demographics, medical history, and diagnoses. This helps researchers expedite the recruitment process, which is often a time-consuming and costly aspect of clinical trials.
The platform also assists in determining trial feasibility by providing insights into patient populations, ensuring trials are designed with realistic expectations. Additionally, Deep 6 AI's solution aids in selecting the most suitable trial sites, factoring in aspects like patient availability, site performance, and investigator experience.
ClinTex
ClinTex is a health technology company founded in 2017 that utilizes blockchain and machine learning to enhance the efficiency and cost-effectiveness of decentralized clinical trials. Their primary product, Clinical Trials Intelligence (CTi), is a suite of seven applications aimed at addressing various clinical trial pain points.
By leveraging blockchain technology, ClinTex ensures data integrity, security, and transparency, which are crucial in clinical trial processes. CTi's machine learning algorithms identify trends, predict outcomes, and optimize trial performance by analyzing data from various sources, enabling proactive issue resolution.
CTi offers a range of applications, including operational analytics, predictive recruitment, and risk-based monitoring, to streamline trial management, reduce patient attrition, and improve trial performance. Its real-time monitoring capabilities allow for better collaboration and informed decision-making among stakeholders.
In summary, ClinTex is revolutionizing decentralized clinical trials with its blockchain-based Clinical Trials Intelligence platform. By utilizing machine learning and blockchain technology, ClinTex aims to improve trial efficiency, reduce costs, and accelerate the development of new medical treatments.
Aural Analytics
Aural Analytics is a health technology company founded in 2015 that focuses on developing speech analytics as a digital biomarker for use in decentralized clinical trials. Their platform is designed to monitor and assess neurological, respiratory, and mental health conditions by analyzing patients' speech patterns.
The technology behind Aural Analytics is built on advanced algorithms and machine learning techniques that extract clinically relevant information from speech data. This includes aspects like pronunciation, fluency, and rhythm, which can provide insights into a patient's health status and disease progression.
One of the key advantages of Aural Analytics' platform is its ability to support multiple languages and work across various devices, making it adaptable and inclusive for clinical trials conducted in different countries and populations.
In decentralized clinical trials, the platform enables remote patient monitoring, reducing the need for in-person assessments. This not only improves patient convenience but also helps in increasing trial participation and retention rates. Additionally, it allows researchers to collect data continuously, providing real-time insights into treatment efficacy and safety.
Savvy Cooperative
Savvy Cooperative is a patient-owned platform founded in 2017, focusing on connecting patients and researchers to enable patient-centric decentralized clinical trials. By facilitating collaboration and data sharing between patients and researchers, Savvy Cooperative aims to improve the clinical trial process and accelerate medical advancements.
As a co-op, Savvy Cooperative empowers patients to become active partners in research, giving them a voice and an opportunity to share their experiences and insights. This approach ensures that trial designs are more patient-centered and reflective of the needs and preferences of the target population. Researchers can access a diverse pool of patient insights, leading to more effective trial outcomes and faster development of new treatments.
Savvy Cooperative offers various services, including patient recruitment, survey design, patient experience research, and user testing. By engaging patients directly, the platform helps address common challenges in clinical trials, such as recruitment and retention, while simultaneously promoting patient advocacy and involvement in the research process.
Teckro
Teckro is a clinical trial technology company founded in 2015 in Ireland. Their digital platform aims to simplify and modernize clinical trial conduct by streamlining trial management and providing mobile access to essential information. Teckro's solution enhances efficiency, communication, and collaboration among sponsors, CROs, trial sites, investigators, and patients.
Key features include mobile access to trial information, real-time updates and notifications, secure communication, machine learning and AI for optimized search and retrieval of trial data, and integration with existing systems. With Teckro's mobile app, investigators and clinical trial staff can quickly access study protocols, inclusion and exclusion criteria, and other critical trial details. The platform's real-time updates help maintain consistency across trial sites and improve overall trial conduct. By leveraging machine learning and artificial intelligence, Teckro ensures efficient decision-making and improved patient care.
myClin
myClin is a Philadelphia-based company that offers a secure, collaborative platform for clinical trial management. Founded in 2008, myClin focuses on streamlining communication, document management, and compliance tracking among clinical research stakeholders, including sponsors, contract research organizations (CROs), clinical trial sites, and investigators.
myClin's platform addresses some of the key challenges in clinical trial management, such as information silos, fragmented communication, and inefficient document handling. By providing a centralized, user-friendly system, myClin makes it easier for clinical trial teams to work together and ensure that essential trial information is easily accessible and up-to-date.
Andaman7
Andaman7 is a Belgium-based health technology company founded in 2015. The company's primary focus is on creating a patient-centered platform for decentralized clinical trials, as well as for the management of personal health records. Their vision is to improve the overall healthcare ecosystem by facilitating collaboration between patients, researchers, and healthcare professionals while ensuring patient privacy and data security.
Andaman7's solution is a mobile application called A7 Health, which serves as a personal health record (PHR) for patients and a remote data collection tool for researchers. The app empowers patients by allowing them to collect, store, and manage their health data securely on their smartphones. Additionally, the platform enables patients to share their data with healthcare providers, researchers, and other authorized parties on a need-to-know basis, maintaining control over their information.
Reify Health
Reify Health is a healthcare technology company that focuses on accelerating clinical trials and improving the overall clinical research process. Founded in 2013 and headquartered in Boston, Massachusetts, Reify Health provides a cloud-based platform designed to streamline clinical trial management for sponsors, clinical research organizations (CROs), and clinical trial sites.
Reify Health's primary product is StudyTeam, a platform that offers an array of tools and features for clinical trial management, and it can be integrated with EHRs.
Examples of AI success in clinical trials: Case study based on IQVIA’s reports
Improved Precision in Identification of Asthma Patients With Unmet Needs Using Predictive Models:
IQVIA's machine learning capabilities allowed the client to improve precision in targeting uncontrolled asthma patients by 30%.
A 6-month analysis of real-world data post-launch showed a 17% reduction in uncontrolled disease status for asthma patients in the region compared to pre-launch figures.
Patients that were accurately predicted to have a high likelihood of having asthma were 86%, eliminating around 30% of non-target patients based on the machine learning prediction versus rules-based findings.
Leveraging Machine Learning to Optimize Care for Patients with Type 2 Diabetes and Obesity:
In a span of just 3 weeks, IQVIA customized two distinct machine learning applications to specifically identify patients with unmet needs in T2D and obesity.
Machine Learning Predicted and Improved Transition of More Patients to Better Treatments:
The machine learning solution expanded patient identification from ~1,000 patients per month to ~2,000 per month.
Patients predicted by IQVIA’s AIML solution to initiate a biologic for asthma had a rate of transition that was 5 times greater than those identified using the previous rules-based approach.
A 3-month analysis of control groups showed that healthcare providers prioritized by IQVIA's machine learning solution had a 20% increase in patients initiating the client’s therapy, thus facilitating quicker access to the right treatment.
Boosting Adherence in MS Patients
Situation: A biotechnology firm wanted to know why multiple sclerosis patients stopped their selective immunosuppressant treatment early.
Solution: Using IQVIA’s Real World Data and machine learning, a predictive model was built. This model used data from IQVIA PharMetrics® Plus and physician data from the American Medical Association.
Outcome: The model predicted patients at high risk of early discontinuation with 81% accuracy, and none with the lowest predicted risk discontinued.
Identifying At-Risk Lymphoma Patients
Situation: A newly FDA-approved drug for lymphoma needed to be promoted as a second line of treatment.
Solution: Using AI/ML, IQVIA identified patients unresponsive to the first line of treatment, allowing more focused promotional efforts.
Outcome: There was a 27% increase in therapy starts for the client’s drug in just 5 months.
Predicting Non-Adherence in Lupus Treatment
Situation: A treatment for lupus showed patients on average received one month less therapy compared to other brands.
Solution: IQVIA employed AI/ML to detect potentially non-adherent patients.
Outcome: About 900 patients at risk of early discontinuation were accurately identified, allowing timely interventions.
Diagnosing Exocrine Pancreatic Insufficiency (EPI)
Situation: A pharma company's product treated EPI, which often goes undiagnosed.
Solution: Machine learning identified potential EPI patients who remained undiagnosed.
Outcome: Over a 20-month period, the model predicted patients were up to 39 times more likely to get an accurate diagnosis and treatment for EPI.
Enhancing Early Treatment Adoption in Oncology
Situation: A client wanted to promote early adoption of their oncology treatment.
Solution: Three machine learning algorithms were developed by IQVIA to identify patients likely to initiate early treatment.
Outcome: The AI algorithm increased the precision of patient identification by 15 times and achieved a treatment transition rate of 28% over three months.
Predicting Treatment Transition in Autoimmune Therapy
Situation: A pharma client had a second-line treatment for an autoimmune disease and faced market competition.
Solution: IQVIA’s machine learning algorithm identified patients likely to seek an alternative treatment, linking them to their healthcare providers.
Outcome: The client was able to prioritize outreach to high-risk patients and physicians, ensuring better treatment transitions.