The Role of AI in Precision Medicine
AI platform RADR® enables precision oncology; the rise of digital pathology (survey); a trend on open-source collaboration (survey); Interview with Dr. James Field (AI+robotics = novel antibodies).
Hi! You are reading ‘Where Tech Meets Bio’, my weekly newsletter about all things ‘tech + bio’: technologies, breakthroughs, and great companies on the cutting edge.
If you've received it, then you either subscribed or someone forwarded it to you. If the latter is the case, subscribe by pressing this button:
Now, let’s get to this week’s topics…
Unveiling Lantern Pharma's Success Story in AI-powered Precision Oncology
(Sponsored by Lantern Pharma)
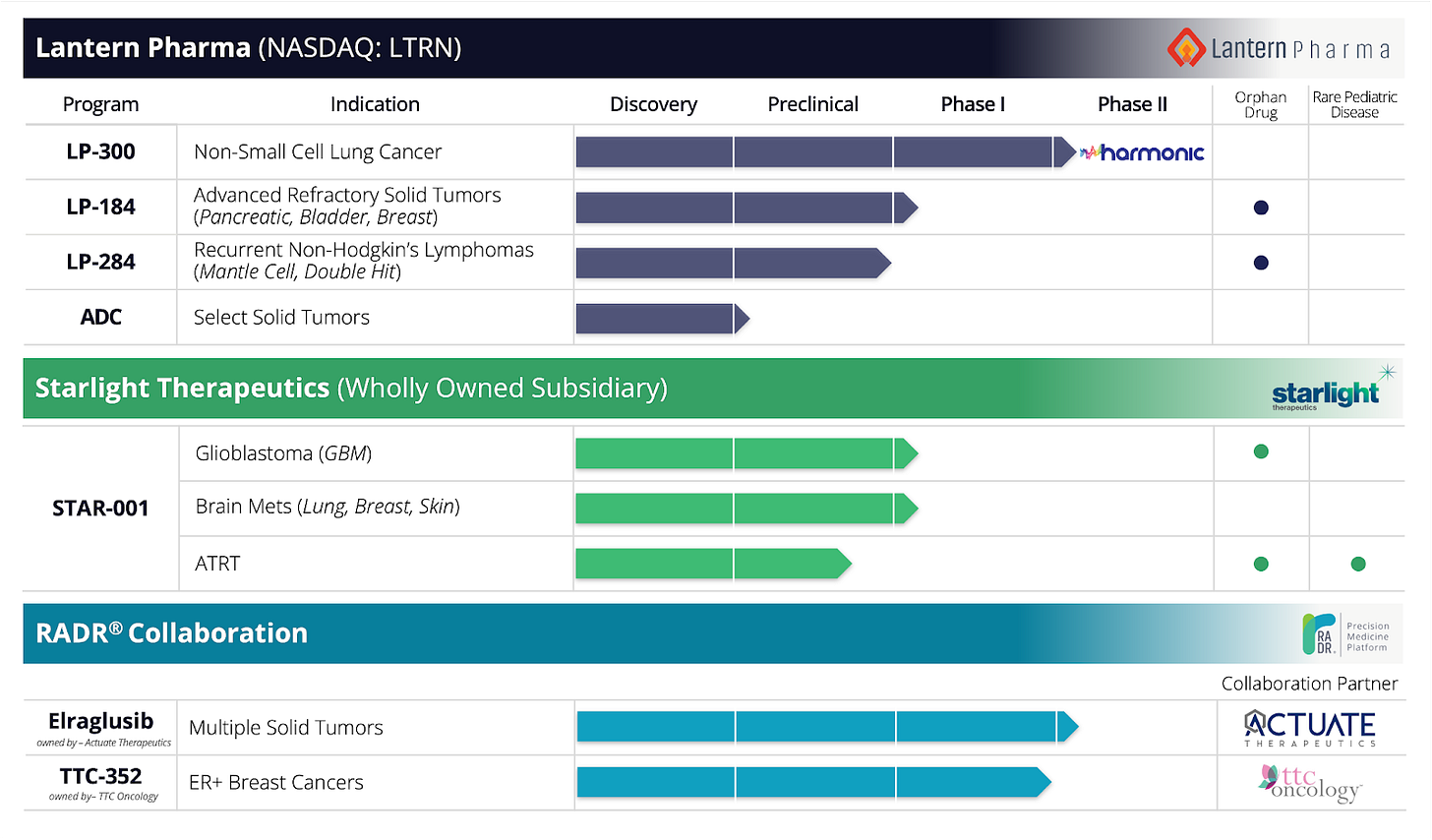
Dallas-based clinical stage biotech Lantern Pharma (NASDAQ: LTRN) has developed AI-driven RADR® platform which is now reimagining oncology drug development by identifying novel mechanism of action (MoA), expanding the space of therapeutic indications and drug combinations, and creating patient response signatures to design focused clinical trials with better success rates.
This is actual precision oncology, enabled by artificial intelligence, so let’s delve into this a bit deeper today.
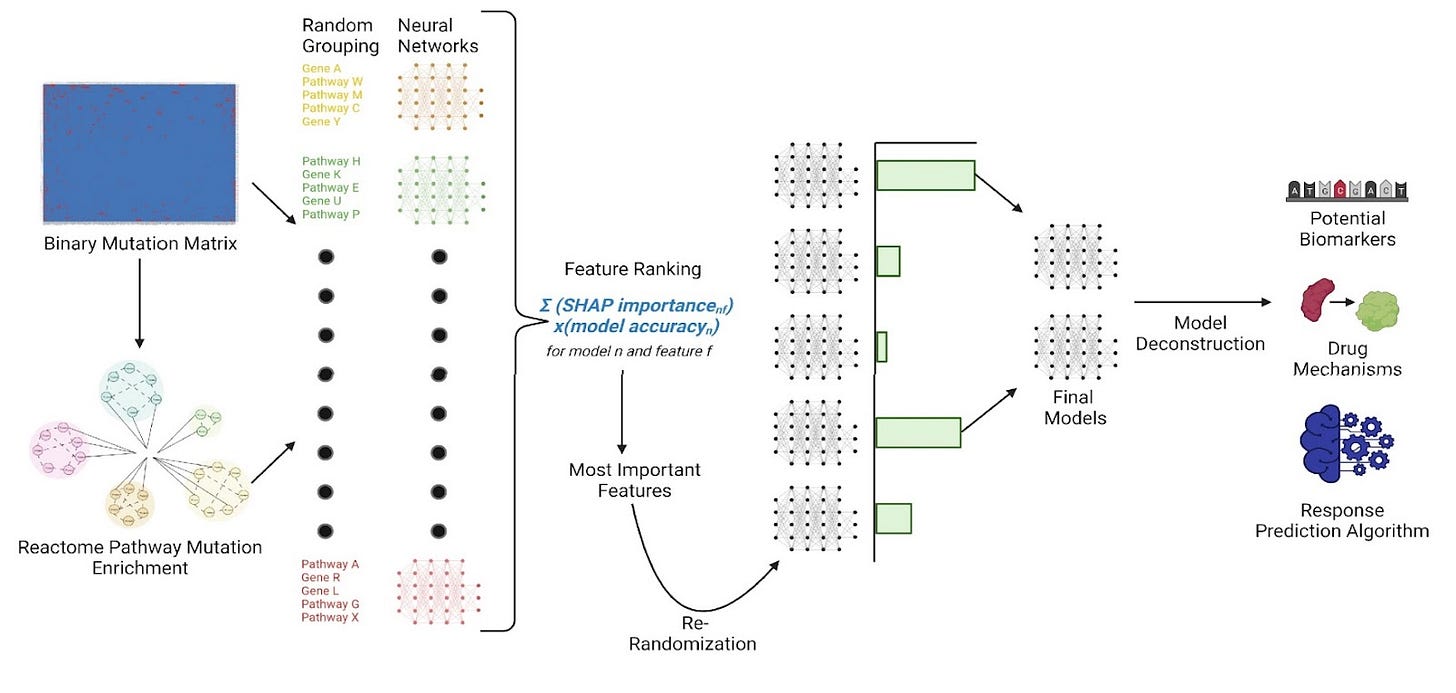
The current challenges in oncology drug discovery include the high failure rate of potential drugs, mischaracterization of drug targets and their inhibitors, and ineffective clinical trial designs. RADR® analyzes over 25 billion data points from genomic and transcriptomic data, thereby helping identify the most suitable patient groups for specific therapies. This increases the likelihood of successful clinical trials and FDA approval, thereby mitigating risks and optimizing success in drug development.
One recent success story is Lantern's IND clearance for LP-184, a synthetically lethal small molecule developed entirely using RADR® insights. The company also dosed its first patient in the Phase 2 multi-center Harmonic™ clinical trial earlier in 2023, while LP-300 increased survival rates for never-smokers with advanced non-small cell lung cancer. Furthermore, Lantern garnered an Orphan Drug Designation (ODD) for LP-284 for treating mantle cell lymphoma, one of the four ODD stage drug candidates in its pipeline.
The operation of Lantern Pharma's RADR® platform involves a complex, yet finely tuned process of data analysis and interpretation.
It begins by ingesting a vast array of data, including transcriptomic, genomic, and drug sensitivity information, sourced from public resources, commercial clinical studies, and Lantern's ex vivo 3D tumor models. Utilizing machine learning algorithms, the platform analyzes over 25 billion data points, 154 drug-tumor interactions, and more than 130,000 patient records across 17 databases. It then correlates this wealth of data with genetic biomarkers and drug activity to create an optimal model predicting drug responses. This unique process assists in generating candidate biomarkers, leading to more precise patient stratification for clinical trial design. Additionally, RADR® can adapt its operational workflow to cater to different stages of drug candidates, aiding in defining indications, combinations, and doses. The platform's capabilities extend to augmenting success rates and de-risking clinical trials, thereby fast-tracking the delivery of vital therapies and potentially improving patient outcomes.
Lantern Pharma welcomes collaborations to help its partners increase the value of their oncology assets.
Lantern Pharma's AI-driven platform holds value for the broader scientific community. The company has a history of successful partnerships with leading organizations, including Actuate Therapeutics (drug candidate Elraglusib, in Phase 2), and TTC Oncology (drug candidate TTC-352, in Phase 1), as well as collaborations with John Hopkins, Fox Chase, UT Health San Antonio, Danish Cancer Society Research Center, Mayo Clinic, and others. For instance, in a recent collaboration with the National Cancer Institute (US), Lantern Pharma utilized its artificial intelligence platform, RADR®, to aid in the discovery of a DNA damaging agent for the ultra-rare cancer known as Atypical Teratoid Rhabdoid Tumors (ATRT), an advancement discussed in a newly published paper.
There is an interesting data from a recent poster (#3079) presented at ASCO 2023, where Lantern Pharma’s RADR® AI platform managed to predict Elraglusib’s clinical response to metastatic melanoma, resistant to checkpoint inhibitors, with 88% accuracy -- for a group of 26 patients, a pretty impressive result as to me.
Read the full article exploring the years-long path of RADR® platform here.
Digital Pathology: The New Frontier in Drug R&D and AI Integration
According to a survey commissioned by Proscia, approximately 75% of life sciences organizations have invested in digital pathology to advance drug R&D, although over half (53%) continue to rely predominantly on legacy software systems. The survey involved 40 senior executives from leading pharmaceutical companies and CROs, revealing a rising necessity for integrating teams, applications, AI, and pathology data through modern software platforms to streamline all R&D stages. It highlighted that digital pathology is perceived as a solution to R&D's major challenges, with 83% of respondents citing improved collaboration and streamlined operations as primary motivations for digitization. Interestingly, the technology is considered by 80% of respondents as a way to bypass lengthy timelines and by 68% as a cost reduction method. Notably, AI implementation in digital pathology is becoming commonplace, with 82% of users having begun to incorporate AI and 87% using image analysis applications. Finally, the survey highlighted that creating data assets through digital pathology, seen as a significant driver by 55% of respondents, offers a new opportunity for valuable real-world data acquisition.
Open-Source Collaboration Pivotal in Life Sciences Research, According to Survey
Seqera Labs' study reveals 72% of 500 surveyed researchers globally regard open-source collaboration as fundamental to their work, having boosted sequencing and avoided duplication. Open-source platforms are used globally, with 41% emphasizing their cross-organizational collaborative potential. International collaboration benefits are recognized by a third of researchers. Community adoption of workflow systems is seen as essential to future research by 41% of respondents, with 34% acknowledging substantial financial savings. Public cloud adoption is increasing, now utilized by 42% of researchers, and up to 80% in the private sector. Amazon Web Services is the preferred provider among potential migrators. These insights underline the transformative potential of open-source tools and cloud services in life sciences research.
Dr. James Field on Breaking the 'Cognition Barrier' in Antibody Discovery with AI and Robotics
Here I am sharing select insights from my latest interview with Dr James Field, CEO @ LabGenius, a London-based biotech which discovers next-generation protein therapeutics using machine learning, robotic automation and synthetic biology.
Andrii: Could you expound on the "cognition barrier" you mention, and elaborate how LabGenius is utilizing mathematical models to understand molecular responses to diseases? What differentiates your approach from conventional methods?
James: Humanity’s incredible success (at least reproductively) can largely be attributed to our capacity to hypothesise and invent. But even this superpower has limits. Concretely, hypothesis-driven innovation requires the inventor to understand the system that they’re working with at an appropriate level of abstraction. Now, here's the rub. There are many arenas in which hypothesis-driven innovation performs poorly because the human brain simply isn't wired to grapple with the complexity of the underlying system.
For example, consider biological systems. Earth’s flora and fauna provide living proof of what can be built with biology, and at the same time highlight the sheer inadequacy of human-led hypothesis-driven innovation within this domain. This shouldn’t come as a surprise. After all, at no point in our evolutionary history has an intuition for manipulating organic matter at the nanoscale conferred a selective advantage!
To address some of humanity’s greatest challenges, it’s clear that we need to break through the ‘cognition barrier’ presented by hypothesis-driven innovation. In the absence of a suitable nootropic, this means that we must develop new forms of innovation that do not require a human to understand the underlying system.
This concept is not new. For decades, scientists, engineers and technologists have dreamt of building ‘robot scientists’ capable of autonomously discovering new knowledge, technologies, and sophisticated real-world products. For protein engineers, that dream is now a real possibility.
The rapid pace of technological development across the fields of synthetic biology, robotic automation, and ML has given us access to the tools required to create a smart, robotic platform capable of intelligently discovering novel therapeutic proteins.
At LabGenius, we’ve spent several years building a platform capable of generating hypotheses, testing them in the lab and then using the resulting data to iteratively refine its understanding of how a molecule’s design determines its performance.
With this approach, we have been able to overcome some major antibody engineering challenges. For example, traditional antibody engineering involves the sequential optimization of different molecular properties through rational design. With this approach, improving one property can inadvertently worsen others. These trade-offs inevitably lead to costly failures or sub-optimal outcomes for patients. In contrast, with our approach, we can now efficiently co-optimise antibodies across multiple features (e.g. potency, efficacy, selectivity, and developability).
Andrii: The results of LabGenius' research indicate a remarkable improvement over clinical benchmarks. Could you discuss the specific methodology employed in accelerating the discovery of these targeted molecules, and how they might alleviate the toxic side effects associated with existing immunotherapies?
James: T-cell engagers (TCEs) are a type of engineered antibody that redirect the immune system's T-cells to recognize and kill cancer cells.
Despite showing real promise, the progression of solid tumour-targeting TCEs through clinical trials has been plagued by issues with dose-limiting ‘on-target, off-tumour’ toxicity. This occurs when healthy cells expressing a tumour-associated antigen (TAA) get unintentionally targeted, which can cause toxic side effects for patients.
In a recent platform demonstration project, we used our ML-driven discovery platform to systematically identify novel TCEs that had strong killing selectivity, were highly potent and also had good developability profiles. The top-performing molecules demonstrated ≥10,000-fold killing selectivity, corresponding to a >400-fold improvement over a relevant clinical benchmark, Runimotamab (a TCE currently in phase I clinical trials) [ref].
The specific form of ML that we used to efficiently navigate design space and find high-performing TCEs is a form of active learning, called Multi-Objective Bayesian Optimization (MOBO).
We're using this capability to develop our own pipeline of highly selective TCEs for the treatment of solid tumours.
Read full interview at BiopharmaTrend.com s week’s topics…