The €25B vs €219B Problem: Europe's Plan to Fix Biotech
Brussels is counting on new laws, sovereign AI, and billions in fresh capital to close the gap. The clock is ticking.
Between 2015 and mid-2025, EU biotech startups attracted €25B in venture capital. In the US, that figure was €219B. To turn things around, Brussels is counting on a legislative package.
Shortly prior to last Christmas the European Commission published a proposal of a European Biotech Act, a strategic initiative aimed at setting up a regulatory framework to strengthen the life sciences sector across the EU. The document has been mostly positively received by the sector leaders as a needed step towards fostering local biotech innovation. Brussels isn’t stopping there. Commission President Ursula von der Leyen pitched EU-Inc, a pan-European company structure meant to solve what many see as the EU’s core startup problem of navigating 27 different bureaucratic regimes. The proposal would let one register in 48 hours, fully online and in English.
The Case for Urgency
Why does it matter? Europe gave the world its first blockbuster pharmaceutical (Aspirin in 1899) and just 30 years ago produced half of all new treatments globally. Today, that share has fallen to just one in five. Even though the EU biotech industry has grown twice as fast as the overall union’s economy over the last decade, it struggles to convert the world’s top science into commercially viable products.
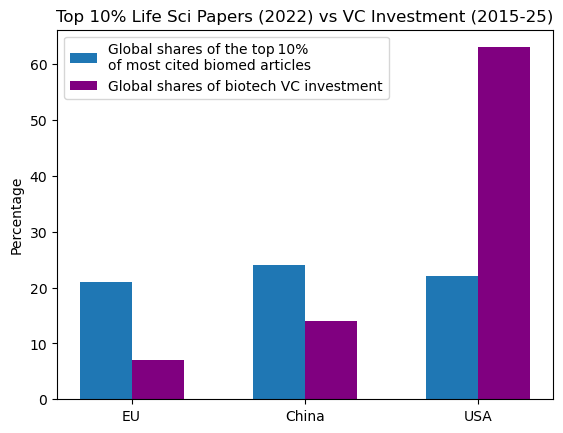
Europe holds a comparable share of the top 10% most-cited biomedical research to the US and China, yet lags significantly behind in venture investment — a gap caused by underdeveloped private equity markets and fragmented, complex regulatory frameworks. The disparity is also visible in listing trends, with 66 of the 67 EU companies that went public over the past six years choosing foreign stock exchanges.
To address this, the Biotech Act includes measures like: