Biologics vs. Small Molecules: Who Wins?
ALSO: This Week's Highlights | Pick of the Week: AI for antibody design | Digging Gems: Drug discovery in space; map of preclinical software; and more...
Hi! I am Andrii Buvailo, and this is my weekly newsletter, ‘Where Tech Meets Bio,’ where I talk about technologies, breakthroughs, and great companies moving the industry forward.
If you've received it, then you either subscribed or someone forwarded it to you. If the latter is the case, subscribe by pressing this button:
Now, let’s get to this week’s topics!
Biologics are forecast to have $120 billion more sales than small molecules by 2027, according to a recent GlobalData report ‘Future of Pharma – Looking Ahead to 2023‘.
Some key insights from the report:
- Biologics are anticipated to represent 55% of all innovative drug sales by 2027.
- Sales growth for biologics is supported by new drug approvals and manufacturing shifts towards these therapies.
- Monoclonal antibodies, like Opdivo, Dupixent, and Keytruda, are major contributors to biologic sales, expected to account for 46% of such sales in 2027.
- Keytruda is projected to represent 4% of all biologics sales in 2027, marketed for various cancer treatments.
- Gene therapies and gene-modified cell therapies are expected to see an increase in sales of over 1,000% from 2022 to 2027.
- Sales for gene therapies are currently driven by pipeline therapies in development, such as RPA-501 by Rocket Pharmaceuticals, which is in Phase I of clinical trials.
Quentin Horgan, Associate Director of Drugs Intelligence at GlobalData, identifies biologics as key drivers of value in the pharmaceutical industry. This, to a great extent, resonates with our views at BiopharmaTrend.
But not entirely. Chemical modalities have been experiencing a kind of renaissance lately. For instance, the progress in artificial intelligence (AI) is accelerating research in chemical modalities too—classic small molecules and more advanced stuff like protein degraders and molecular glues.
What are the differences between the modalities, in a nutshell? I tried to summarize key attributes in a table below, illustrating challenges and opportunities available in both “camps.”
Color keys: gray (no apparent advantage in neither camp); green (an advantageous situation); yellow (a disadvantageous situation).
(You can open the image in the new tab to zoom in as needed):
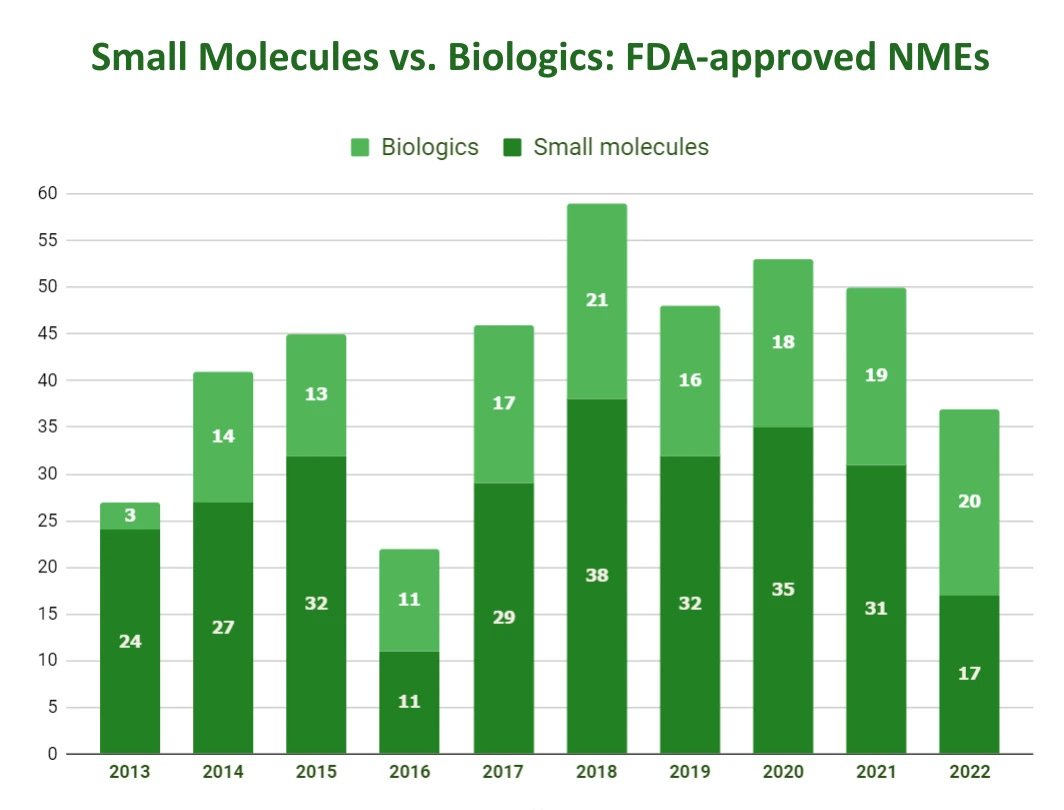
If we talk about the statistics of FDA approvals, I would say biologics are slowly outgrowing SMs in numbers per year, with 2022 being the first year with more approvals in biologics vs. small molecules.
While it is hard to surprise anyone saying that the drug discovery of biologics is clearly growing in the diversity of modalities and overall interest and attention from investors and corporations, it is fair to say that technological progress over the past decade led to a serious upgrade in the arsenal of medicinal chemists too.
A new article in C&EN nicely summarizes the key innovative areas where chemical modalities are shining now. These are changing the way we create treatments, moving away from the old method of fitting a drug to target proteins like a key in a lock, to more complex ways of tackling illnesses.
Covalent inhibitors
The tide turned with covalent inhibitors' debut, which bind irreversibly to proteins, sidestepping the need for a suitable protein pocket. This leap was demonstrated by Kevan Shokat’s team at the University of California, San Francisco, with their groundbreaking work on a KRAS mutant, pivotal in cancer progression.
PROTACs
Then, the proteolysis-targeting chimeras (PROTACs) developed by Craig Crews’s laboratory at Yale University, opened a novel therapeutic avenue. These bifunctional molecules, although yet to clear the final regulatory checks, are lauded for their ability to mark disease-causing proteins for cellular degradation. Arvinas, leading in this field with structures like Bavdegalutamide and Vepdegestrant, has pioneered PROTACs, underscoring the potential of induced proximity drugs.
Molecular ‘glues’
Similarly transformative are the molecular glues, a more diminutive approach to induce protein degradation. Companies like Proxygen and Monte Rosa Therapeutics have seized this technology, striking deals with pharmaceutical giants such as Merck KGaA, Merck & Co., and Roche to advance glues against seemingly undruggable targets in cancer and neurological diseases.
Macrocycles
Not to be overlooked is the advent of macrocycles and beyond-rule-of-5 molecules. These larger constructs defy the traditional small-molecule framework, offering new methods of disease engagement. Cyclosporin A exemplifies such a macrocycle, an immunosuppressant that has been in use since long before the term 'beyond-rule-of-5' was coined.
Small molecules targeting RNA
Venturing beyond proteins, the domain of RNA has unfolded as a new frontier for small-molecule intervention. Here, companies like Arrakis Therapeutics, co-founded by chemist Jennifer Petter, are making strides with modalities designed to bind, modify, or even degrade RNA, expanding the arsenal against genetic and molecular disorders.
RNA was long considered undruggable by small molecules, but things changed. Under the guidance of chemists like Matthew Disney, Expansion Therapeutics is another example of a company pushing the envelope by developing molecules that engage with RNA structures.
Weekly Highlights
How Biden's AI executive order could impact pharma, biotech
President Biden has enacted an expansive executive order to regulate artificial intelligence, setting a precedent for future U.S. government policies. Highlighted by MIT's Sinan Aral as a major move in the absence of Congressional action, the order sets guidelines for AI's ethical use, ensuring safety, privacy, and equity. It tasks NIST with establishing robust standards and includes provisions for worker protection and maintaining competitiveness. Notably, the directive could influence the operations of biotech and pharma companies that deploy AI for drug discovery and development by demanding higher safety and privacy standards. It also calls for bipartisan legislation on data protection, which could reshape data handling across various industries.
DeepMind’s latest AlphaFold model is more useful for drug discovery
The new AlphaFold models by Isomorphic Labs mark an evolution in AI's role in drug discovery, offering a substantial improvement in predicting how drugs interact with proteins. This progress addresses previous limitations where AlphaFold's predictions did not effectively translate to drug development due to its training on protein structures alone. A key breakthrough is that these models were trained on protein-ligand complexes, enhancing their accuracy in molecular docking benchmarks, and demonstrating the importance of targeted training data.
Key takeaway: The effectiveness of AI in biology hinges on the specificity of its training data; to accurately model complex biological interactions for drug discovery, AI must be trained on detailed, relevant biological complexes.
BioAge is set to launch a Phase 2 clinical trial in mid-2024 to test the efficacy of azelaprag, an oral apelin receptor agonist, combined with Eli Lilly's tirzepatide, in treating obesity. This collaboration benefits from Lilly's expertise in trial design and drug development, with the objective of achieving significant healthy weight loss through an all-oral medication regimen. Preclinical studies show that this drug combination not only enhances weight loss but also improves body composition and blood glucose levels, aligning with BioAge's aim to prolong healthy lifespans.
Mimics human tissue, fights bacteria: new biomaterial hits the sweet spot
Scientists at UNSW Sydney have developed a new hydrogel called 'Trpzip' that mimics human tissue and offers antimicrobial properties, potentially reducing the need for animal-derived materials in biomedical research and applications. This self-assembling, self-healing material is suitable for 3D bioprinting and injectable medical applications due to its resilience and the ability to reform after being deformed. The discovery, which has implications for medicine, food, and manufacturing technology, could lead to more ethical and uniform alternatives to natural hydrogels currently used across various industries.
DNA-encoded library-enabled discovery of proximity-inducing small molecules
The study utilized DNA-encoded libraries to create a vast array of compounds that could connect the E3 ligase VHL protein to various bromodomains, with the goal of identifying molecules capable of forcing these proteins to interact. They succeeded in both the high-throughput identification of these compounds and in demonstrating their ability to induce protein-protein interactions, as evidenced by the structural analysis of a resulting ternary complex.
Pick of the Week
AI is transforming the field of antibody discovery, but the key to success is the business model.
I’ve stumbled upon a quite interesting article in Genengnews about California-based BigHat Biosciences.
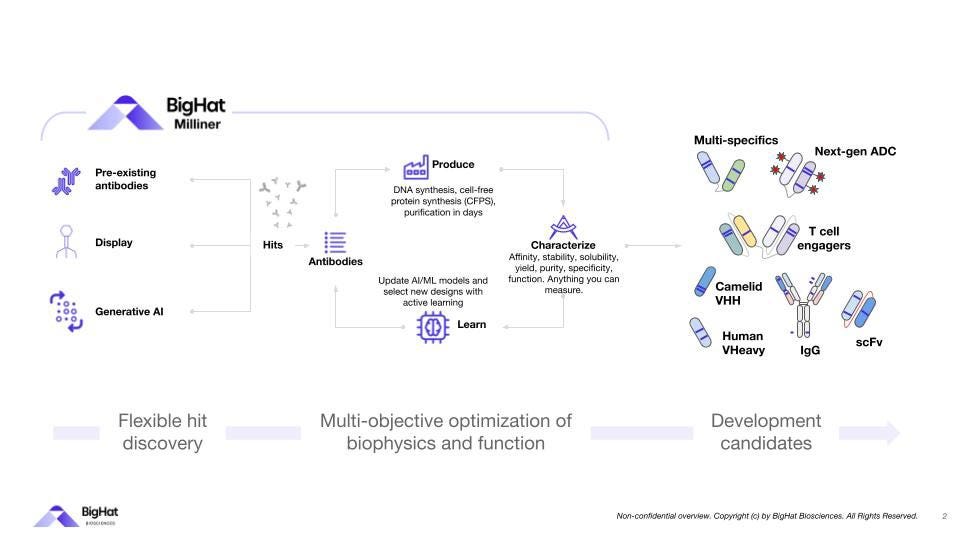
BigHat uses its platform, Milliner, to integrate advanced machine learning with synthetic biology to make Abs development faster and more successful.
The technology enables rapid design and characterization of a diverse array of next-gen therapeutic Abs, such as Bi-specific T-cell engagers (BiTEs), Bispecific antibodies (BsAbs), single-chain variable fragments (scFvs), and antibody-drug conjugates (ADCs).
An interesting aspect of BigHat is its capability to model and optimize single-domain antibodies (nanobodies), like VHH from camelids. Those are ~15 kD, which is way smaller than human IgG mAbs, providing targeted antigen recognition with high affinity while ensuring thermostability.
While BigHat’s pipeline is still preclinical, what I find really interesting is its business model. They seem to have the two key elements for success: a validated AI platform and a high-throughput wet lab for generating lots of data.
They emphasize collaborations in areas with well-understood biology and high-quality assays that are predictive of clinical efficacy and safety. This focus enables the company to refine their molecules directly against these measurements, positioning themselves in fields where traditional monoclonal antibodies are established but where AI/ML can push into new molecular spaces.
The company cultivates a few deep, value-oriented partnerships rather than spreading efforts thinly. Their internal pipeline focuses on oncology and inflammation—sectors ripe with unmet needs that are difficult to address with conventional protein engineering.
On the collaboration front, BigHat has struck significant partnerships, like the one with Merck, to tackle complex antibody design issues using their Milliner platform. Their work with Amgen has already hit key milestones, showcasing their platform’s efficiency in optimizing single-domain antibodies.
2022 acquisition of Frugi Biotechnology enhances their capabilities in rapid, cost-effective antibody synthesis, which is central to BigHat's innovative edge.
Financially, BigHat is robust, having raised over $100 million, with a significant $80 million Series B funding round led by Section 32 and supported by a combination of new and returning investors.
I am really curious to see their biologics in clinical trials and how they would perform. It is, of course, hard to say. But what I like about this business model is its flexibility.
Digging Gems
Drug development in space is a big deal!
Florida-based Redwire Space (NYSE: RDW), a space infrastructure company, has just announced the first spaceflight mission for its cutting-edge in-space pharmaceutical manufacturing platform, PIL-BOX.
This first mission sends Eli Lilly and Company’s materials to space to conduct three critical experiments in microgravity, focused on developing advanced treatments for diabetes, cardiovascular disease, and pain.
Understanding crystal growth and design can inform the entire drug development and design process as pharmaceutical companies look to deliver new, optimized treatments to help patients on Earth. Previous spaceflight investigations indicate that growing crystals in the microgravity of space could yield a more uniform product with fewer imperfections.
The new platform by Redwire will launch onboard SpaceX’s 29th cargo resupply services mission (SpaceX-29) for NASA to the International Space Station (ISS).
Back in March, Bristol Myers Squibb embarked on its second mission to research protein crystallization in microgravity aboard the ISS, aiming to develop more stable and concentrated biologic medicines that could lead to simpler, at-home patient treatments. BMS experiment was launched as part of SpaceX's 27th commercial resupply mission to the ISS.
Also, Merck has been working on crystalline suspensions of monoclonal antibodies, like those in Keytruda, their oncology drug, finding that crystals grown in space have lower viscosity and better injectability properties. This could transform the drug delivery method from lengthy infusions to quick injections.
Protein crystallization is not the only interest of life science companies in space.
On SpaceX-29, Redwire is also launching materials for an investigation that will bioprint cardiac tissue on orbit using Redwire’s BioFabrication Facility (BFF). This type of technology could be used to develop heart patches that can be applied to the outside of damaged hearts and advance our ability to print complex, thick tissues that cannot be produced on Earth. In September, Redwire announced that it had successfully 3D-bioprinted the first human knee meniscus on orbit using BFF. The print was returned to Earth for further study and analysis.
This week, I came across this media project S³ by Jason Carman where the guy creates inspiring videos about startups, including biotechs. They are amazing!
Make sure to check Episode 16 of S³ features Amber Bio who are pioneering new gene editing modalities.
Folks at Deep Origin built the life science software landscape, a map of more than 300 software products and tools in preclinical R&D, organized by function and specialty (e.g., drug modality, data type). Increadible effort!